Loading AI tools
居禮點(英語:Curie point),又作居禮溫度(Curie temperature,Tc)或磁性轉變點。是指磁性材料中自發磁化強度降到零時的溫度,是鐵磁性或亞鐵磁性物質轉變成順磁性物質的臨界點。低於居禮點溫度時該物質成為鐵磁體,此時和材料有關的磁場很難改變。當溫度高於居禮點時,該物質成為順磁體,磁體的磁場很容易隨周圍磁場的改變而改變。這時的磁敏感度約為10-6。居禮點由物質的化學成分和晶體結構決定。居禮溫度是以皮埃爾·居禮命名的,他表明在臨界溫度下磁性材料會失去磁性。
居禮點的溫度可以用平均場理論估計。
| 材料 | 居禮 溫度 (K) |
|---|---|
| 鐵 (Fe) | 1043 |
| 鈷 (Co) | 1400 |
| 鎳 (Ni) | 627 |
| 釓 (Gd) | 292 |
| 鏑 (Dy) | 88 |
| 鉍化錳 (MnBi) | 630 |
| 銻化錳(MnSb) | 587 |
| 二氧化鉻 (CrO2) | 386 |
| 砷化錳 | 318 |
| 氧化銪 | 69 |
| 氧化鐵 (Fe2O3) | 948 |
| 四氧化三鐵 (FeOFe2O3) | 858 |
| 氧化鎳-氧化鐵NiO–Fe2O3 | 858 |
| 氧化銅-氧化鐵CuO–Fe2O3 | 728 |
| 氧化鎂-氧化鐵MgO–Fe2O3 | 713 |
| 氧化錳-氧化鐵MnO–Fe2O3 | 573 |
| 釔鐵石榴石 (Y3Fe5O12) | 560 |
| 釹磁鐵 | 583—673 |
| 鋁鎳鈷合金 | 973—1133 |
| 釤鈷磁鐵 | 993—1073 |
| 鍶鐵氧體 | 723 |
磁矩是原子內的永久偶極矩,包含電子的角動量和自旋[4],他們之間的關係是 , me 是電子質量, μl 是磁矩, l是角動量; 這個比例被稱作 gyromagnetic ratio(旋磁比).
原子中的電子從它們自己的角動量和它們圍繞原子核的軌域動量貢獻磁矩。與來自電子的磁矩相比,來自原子核的磁矩是微不足道的。[5] 熱作用在更高能量的電子上結果就是擾亂了秩序,並破壞了偶極子之間的對齊。
鐵磁性、順磁性、亞鐵磁性和反鐵磁性材料有不同的固有磁矩結構。在材料特定的居禮溫度(TC)下,這些屬性會發生變化。從反鐵磁性到順磁性(或反之亦然)的過渡發生在奈爾溫度(TN), 這與居禮溫度類似。
鐵磁性,順磁性,亞鐵磁性和反鐵磁性結構由固有磁矩組成。 如果結構中的所有電子都配對,則由於它們的相反自旋和角動量,這些力矩會抵消。 因此,即使施加磁場,這些材料也具有不同的性質,並且沒有居禮溫度。[6][7]
當一些材料的溫度高於居禮點時,材料會表現出順磁性,這樣的材料叫順磁性材料。當沒有受到外部磁場的影響時,順磁性材料不會表現磁性;反之則會表現磁性。沒有受到外部磁場影響時,材料內部的磁矩是無序排列的。也就是說,材料內部的粒子不整齊且沒有順磁力線方向排列。當受到磁場影響時,這些磁矩會順磁場線整齊排列[8][9][10],並且產生感應磁場[10][11]。
對於順磁性,這種對外加磁場的響應是正的,稱為磁化率。[6] 磁化率僅適用於居禮溫度以上的無序狀態。[12]
順磁性的來源(具有居禮溫度的材料)包括:[13]
- 所有含未配對電子的原子;
- 內電子層未被填滿的原子;
- 自由基;
- 金屬。
超過居禮溫度後,原子被激發, 旋轉的方向變成隨機的[7] ,這種方向可以被作用場重新調整,此時即變為順磁性。在居禮溫度以下,材料的固有結構經歷了一次相變,[14] 原子變為有序,材料具有鐵磁性。[10] 與鐵磁性材料的磁場相比,順磁性材料的感應磁場非常弱。[14]
材料僅在其相應的居禮溫度以下具有鐵磁性。在沒有外加磁場的情況下,鐵磁材料具有磁性。
當沒有外加磁場時,材料具有自發磁化,這是有序磁矩的結果;也就是說,對於鐵磁性材料,原子具有某種對稱性並且在同一方向上排列,從而產生永久磁場。
磁性相互作用通過交換相互作用結合在一起;否則,熱無序將克服磁矩的弱相互作用。交換相互作用的平行電子占據同一時間點的可能性為零,這意味著材料中會有一個傾向的平行排列。[15] 在這個過程中,玻爾茲曼因子貢獻很大,因為它傾向於使相互作用的粒子在同一方向上排列。[16] 這會導致鐵磁體具有較強的磁場和較高的居禮溫度,約 1000K(730℃)[17]
在居禮溫度以下,原子有序排列,從而導致自發磁性,材料具有鐵磁性。在居禮溫度以上,該材料是順磁性的,因為當該材料經歷相變時,原子會失去其有序的磁矩。[14]
材料僅在其相應的居禮溫度以下具有亞鐵磁性。在沒有外加磁場的情況下,亞鐵磁材料具有磁性,並由兩種不同離子組成。[18]
當沒有外加磁場時,材料具有自發磁化,這是有序磁矩的結果;也就是說,對於亞鐵磁性材料,一種離子的磁矩對準一個方向,有一個大小,另一種離子的磁矩對準相反方向,有一個不同的大小。因為磁矩在相反的方向有著不同的大小,所以仍然有自發磁化,存在磁場。[18]
和鐵磁性材料相似,磁性相互作用通過交換相互作用結合在一起。但是,磁矩的方向是反平行的,導致淨勢是一個減另一個。[18]
低於居禮點時,每個離子的原子都反平行對齊,有著不同的磁矩,造成自發磁化;材料具有亞鐵磁性。高於居禮點時,該材料是順磁性的,因為當該材料經歷相變時,原子會失去其有序的磁矩。[18]
材料僅在其相應的奈爾溫度以下具有反鐵磁性。這與居禮溫度相似,高於奈爾溫度時,該材料經歷相變,變成順磁性。也就是說,熱能變得足夠大,足以破壞材料內的微觀磁有序性。 [19]它以路易·奈爾(Louis Néel,1904-2000 年)的名字命名,他因在該領域的工作而獲得了 1970 年的諾貝爾物理學獎。
材料有方向相反的相等磁矩,導致在奈爾溫度以下磁矩為零和淨磁性為零。反鐵磁性材料在有或沒有外加磁場的情況下有很弱的磁性。
與鐵磁性材料相似,磁性相互作用通過交換相互作用結合在一起;否則,熱無序將克服磁矩的弱相互作用。[15][20]奈爾溫度時無序出現。[20]
下面列表中有幾種物質的奈爾溫度:[21]
| Definition | |
|---|---|
| χ | the magnetic susceptibility; the influence of an applied magnetic field on a material |
| M | the magnetic moments per unit volume |
| H | the macroscopic magnetic field |
| B | the magnetic field |
| C | the material-specific Curie constant |
| Below TC | Above TC[47] |
|---|---|
| Ferroelectric | ↔ Dielectric (paraelectric) |
| Antiferroelectric | ↔ Dielectric (paraelectric) |
| Ferrielectric | ↔ Dielectric (paraelectric) |
| Helielectric | ↔ Dielectric (paraelectric) |
已隱藏部分未翻譯內容,歡迎參與翻譯。
居禮 - 韋斯定律
居禮-韋斯定律是居禮定律的修正版本,是基於平均場論近似的簡單模型,在材料溫度T遠高於其對應居禮溫度TC(即T ≫ TC)時較為適用,但卻因原子間的局部波動作用無法在居禮點附近對磁化率χ進行描述[23]。而T < TC的情況下,在居禮定律和居禮-韋斯定律均不成立。
Curie's law for a paramagnetic material:[24]
| Avagadro常數 | |
| µ0 | the permeability of free space. Note: in CGS units is taken to equal one.[26] |
| g | the Landé g-factor |
| J(J + 1) | the eigenvalue for eigenstate J2 for the stationary states within the incomplete atoms shells (electrons unpaired) |
| µB | the Bohr Magneton |
| kB | Boltzmann's constant |
| total magnetism | is N number of magnetic moments per unit volume |
The Curie–Weiss law is then derived from Curie's law to be:
where:
For full derivation see 居禮-韋斯定律.
物理
從上方接近居禮溫度
As the Curie–Weiss law is an approximation, a more accurate model is needed when the temperature, T, approaches the material's Curie temperature, TC.
Magnetic susceptibility occurs above the Curie temperature.
An accurate model of critical behaviour for magnetic susceptibility with critical exponent γ:
The critical exponent differs between materials and for the mean-field model is taken as γ = 1.[28]
As temperature is inversely proportional to magnetic susceptibility, when T approaches TC the denominator tends to zero and the magnetic susceptibility approaches infinity allowing magnetism to occur. This is a spontaneous magnetism which is a property of ferromagnetic and ferrimagnetic materials.[29][30]
從下方接近居禮溫度
Magnetism depends on temperature and spontaneous magnetism occurs below the Curie temperature. An accurate model of critical behaviour for spontaneous magnetism with critical exponent β:
The critical exponent differs between materials and for the mean-field model as taken as β = 1/2 where T ≪ TC.[28]
The spontaneous magnetism approaches zero as the temperature increases towards the materials Curie temperature.
接近絕對零度(0開爾文)
The spontaneous magnetism, occurring in ferromagnetic, ferrimagnetic and antiferromagnetic materials, approaches zero as the temperature increases towards the material's Curie temperature. Spontaneous magnetism is at its maximum as the temperature approaches 0 K.[31] That is, the magnetic moments are completely aligned and at their strongest magnitude of magnetism due to no thermal disturbance.
In paramagnetic materials temperature is sufficient to overcome the ordered alignments. As the temperature approaches 0 K, the 熵 decreases to zero, that is, the disorder decreases and becomes ordered. This occurs without the presence of an applied magnetic field and obeys the 熱力學第三定律.[15]
Both Curie's law and the Curie–Weiss law fail as the temperature approaches 0 K. This is because they depend on the magnetic susceptibility which only applies when the state is disordered.[32]
硫酸釓 continues to satisfy Curie's law at 1 K. Between 0 and 1 K the law fails to hold and a sudden change in the intrinsic structure occurs at the Curie temperature.[33]
Ising相變模型
The Ising model is mathematically based and can analyse the critical points of phase transitions in ferromagnetic order due to spins of electrons having magnitudes of ±1/2. The spins interact with their neighbouring dipole electrons in the structure and here the Ising model can predict their behaviour with each other.[34][35]
This model is important for solving and understanding the concepts of phase transitions and hence solving the Curie temperature. As a result, many different dependencies that affect the Curie temperature can be analysed.
For example, the surface and bulk properties depend on the alignment and magnitude of spins and the Ising model can determine the effects of magnetism in this system.
Weiss磁疇和表面和體積居禮溫度
Materials structures consist of intrinsic magnetic moments which are separated into domains called Weiss domains.[36] This can result in ferromagnetic materials having no spontaneous magnetism as domains could potentially balance each other out.[36] The position of particles can therefore have different orientations around the surface than the main part (bulk) of the material. This property directly affects the Curie temperature as there can be a bulk Curie temperature TB and a different surface Curie temperature TS for a material.[37]
This allows for the surface Curie temperature to be ferromagnetic above the bulk Curie temperature when the main state is disordered, i.e. Ordered and disordered states occur simultaneously.[34]
The surface and bulk properties can be predicted by the Ising model and electron capture spectroscopy can be used to detect the electron spins and hence the magnetic moments on the surface of the material. An average total magnetism is taken from the bulk and surface temperatures to calculate the Curie temperature from the material, noting the bulk contributes more.[34][38]
The angular momentum of an electron is either +ħ/2 or −ħ/2 due to it having a spin of 1/2, which gives a specific size of magnetic moment to the electron; the Bohr magneton.[39] Electrons orbiting around the nucleus in a current loop create a magnetic field which depends on the Bohr Magneton and magnetic quantum number.[39] Therefore, the magnetic moments are related between angular and orbital momentum and affect each other. Angular momentum contributes twice as much to magnetic moments than orbital.[40]
For terbium which is a rare-earth metal and has a high orbital angular momentum the magnetic moment is strong enough to affect the order above its bulk temperatures. It is said to have a high anisotropy on the surface, that is it is highly directed in one orientation. It remains ferromagnetic on its surface above its Curie temperature while its bulk becomes ferrimagnetic and then at higher temperatures its surface remains ferrimagnetic above its bulk Néel Temperature before becoming completely disordered and paramagnetic with increasing temperature. The anisotropy in the bulk is different from its surface anisotropy just above these phase changes as the magnetic moments will be ordered differently or ordered in paramagnetic materials.[37]
更改材料的居禮溫度
Composite materials
Composite materials, that is, materials composed from other materials with different properties, can change the Curie temperature. For example, a composite which has silver in it can create spaces for oxygen molecules in bonding which decreases the Curie temperature[41] as the crystal lattice will not be as compact.
The alignment of magnetic moments in the composite material affects the Curie temperature. If the materials moments are parallel with each other the Curie temperature will increase and if perpendicular the Curie temperature will decrease[41] as either more or less thermal energy will be needed to destroy the alignments.
Preparing composite materials through different temperatures can result in different final compositions which will have different Curie temperatures.[42] Doping a material can also affect its Curie temperature.[42]
The density of nanocomposite materials changes the Curie temperature. Nanocomposites are compact structures on a nano-scale. The structure is built up of high and low bulk Curie temperatures, however will only have one mean-field Curie temperature. A higher density of lower bulk temperatures results in a lower mean-field Curie temperature and a higher density of higher bulk temperature significantly increases the mean-field Curie temperature. In more than one dimension the Curie temperature begins to increase as the magnetic moments will need more thermal energy to overcome the ordered structure.[38]
Particle size
The size of particles in a material's crystal lattice changes the Curie temperature. Due to the small size of particles (nanoparticles) the fluctuations of electron spins become more prominent, this results in the Curie temperature drastically decreasing when the size of particles decrease as the fluctuations cause disorder. The size of a particle also affects the anisotropy causing alignment to become less stable and thus lead to disorder in magnetic moments.[34][43]
The extreme of this is superparamagnetism which only occurs in small ferromagnetic particles and is where fluctuations are very influential causing magnetic moments to change direction randomly and thus create disorder.
The Curie temperature of nanoparticles are also affected by the crystal lattice structure, body-centred cubic (bcc), face-centred cubic (fcc) and a hexagonal structure (hcp) all have different Curie temperatures due to magnetic moments reacting to their neighbouring electron spins. fcc and hcp have tighter structures and as a results have higher Curie temperatures than bcc as the magnetic moments have stronger effects when closer together.[34] This is known as the coordination number which is the number of nearest neighbouring particles in a structure. This indicates a lower coordination number at the surface of a material than the bulk which leads to the surface becoming less significant when the temperature is approaching the Curie temperature. In smaller systems the coordination number for the surface is more significant and the magnetic moments have a stronger affect on the system.[34]
Although fluctuations in particles can be minuscule, they are heavily dependent on the structure of crystal lattices as they react with their nearest neighbouring particles. Fluctuations are also affected by the exchange interaction[43] as parallel facing magnetic moments are favoured and therefore have less disturbance and disorder, therefore a tighter structure influences a stronger magnetism and therefore a higher Curie temperature.
Pressure
Pressure changes a material's Curie temperature. Increasing pressure on the crystal lattice decreases the volume of the system. Pressure directly affects the kinetic energy in particles as movement increases causing the vibrations to disrupt the order of magnetic moments. This is similar to temperature as it also increases the kinetic energy of particles and destroys the order of magnetic moments and magnetism.[44]
Pressure also affects the density of states (DOS).[44] Here the DOS decreases causing the number of electrons available to the system to decrease. This leads to the number of magnetic moments decreasing as they depend on electron spins. It would be expected because of this that the Curie temperature would decrease however it increases. This is the result of the exchange interaction. The exchange interaction favours the aligned parallel magnetic moments due to electrons being unable to occupy the same space in time[15] and as this is increased due to the volume decreasing the Curie temperature increases with pressure. The Curie temperature is made up of a combination of dependencies on kinetic energy and the DOS.[44]
The concentration of particles also affects the Curie temperature when pressure is being applied and can result in a decrease in Curie temperature when the concentration is above a certain percent.[44]
Orbital ordering
Orbital ordering changes the Curie temperature of a material. Orbital ordering can be controlled through applied strains.[45] This is a function that determines the wave of a single electron or paired electrons inside the material. Having control over the probability of where the electron will be allows the Curie temperature to be altered. For example, the delocalised electrons can be moved onto the same plane by applied strains within the crystal lattice.[45]
The Curie temperature is seen to increase greatly due to electrons being packed together in the same plane, they are forced to align due to the exchange interaction and thus increases the strength of the magnetic moments which prevents thermal disorder at lower temperatures.
鐵電材料中的居禮溫度
In analogy to ferromagnetic and paramagnetic materials, the term Curie temperature (TC) is also applied to the temperature at which a ferroelectric material transitions to being paraelectric. Hence, TC is the temperature where ferroelectric materials lose their spontaneous polarisation as a first or second order phase change occurs. In case of a second order transition the Curie Weiss temperature T0 which defines the maximum of the dielectric constant is equal to the Curie temperature. However, the Curie temperature can be 10 K higher than T0 in case of a first order transition.[46]
Ferroelectric and dielectric
Materials are only ferroelectric below their corresponding transition temperature T0.[48] Ferroelectric materials are all pyroelectric and therefore have a spontaneous electric polarisation as the structures are unsymmetrical.
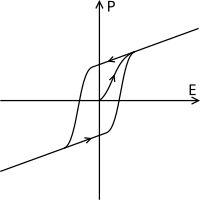
Ferroelectric materials' polarization is subject to hysteresis (Figure 4); that is they are dependent on their past state as well as their current state. As an electric field is applied the dipoles are forced to align and polarisation is created, when the electric field is removed polarisation remains. The hysteresis loop depends on temperature and as a result as the temperature is increased and reaches T0 the two curves become one curve as shown in the dielectric polarisation (Figure 5).[49]
相對介電常數
A modified version of the Curie–Weiss law applies to the dielectric constant, also known as the relative permittivity:[46][50]
應用
A heat-induced ferromagnetic-paramagnetic transition is used in magneto-optical storage media, for erasing and writing of new data. Famous examples include the Sony Minidisc format, as well as the now-obsolete CD-MO format. Curie point electro-magnets have been proposed and tested for actuation mechanisms in passive safety systems of fast breeder reactors, where control rods are dropped into the reactor core if the actuation mechanism heats up beyond the material's curie point.[51] Other uses include temperature control in soldering irons,[52] and stabilizing the magnetic field of tachometer generators against temperature variation.[53]
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.