阿昔洛韦
抗病毒药物 来自维基百科,自由的百科全书
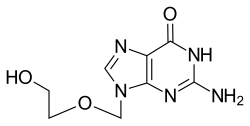
阿昔洛韦 (INN:aciclovir,USAN:acyclovir,ACV)[3]又称无环鸟苷[4],是一种化学合成的类核苷抗病毒药物[5],主要用于治疗单纯疱疹病毒(HSV)、水痘和带状疱疹的感染[6]。其他用途包括预防器官移植后巨细胞病毒属感染,和预防人类疱疹病毒第四型(EBV)感染之后的严重并发症[6][7]。它有口服给药、涂抹用乳霜或静脉注射的形式[6]。
 | |
 | |
| 临床资料 | |
|---|---|
| 读音 | /eɪˈsaɪkloʊvɪər/ |
| 商品名 | Zovirax及其他[1] |
| 其他名称 | Acycloguanosine, acyclovir (BAN UK), acyclovir (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a681045 |
| 核准状况 | |
| 怀孕分级 |
|
| 给药途径 | 静脉注射, 口服给药, 外用药物, 眼部给药 |
| ATC码 | |
| 法律规范状态 | |
| 法律规范 |
|
| 药物动力学数据 | |
| 生物利用度 | 15–20% (口服)[2] |
| 血浆蛋白结合率 | 9–33%[2] |
| 药物代谢 | 肝脏 |
| 生物半衰期 | 2–4小时 |
| 排泄途径 | 肾脏 (62–90%保持原形式) |
| 识别信息 | |
| |
| CAS号 | 59277-89-3 |
| PubChem CID | |
| PubChem SID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB配体ID | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.056.059 |
| 化学信息 | |
| 化学式 | C8H11N5O3 |
| 摩尔质量 | 225.21 g·mol−1 |
| 3D模型(JSmol) | |
| 熔点 | 256.5 °C(493.7 °F) |
| |
| |
使用后常见的副作用有恶心和腹泻。[6]可能的的严重副作用有肾脏问题和血小板减少。[6]肝脏或肾脏功能较差的人使用时,应更为谨慎。[6]一般认为个体在怀孕期间使用对胎儿属于安全,尚未观察到任何危害案例。[6][8]使用个体进行母乳哺育,对于婴儿似乎属于安全。[9][10]阿昔洛韦是一种模仿鸟苷的核苷类似物,[6]其作用是抑制病毒生产DNA的能力。[6]
阿昔洛韦于1974年由葛兰素史克药业取得美国专利,并于1981年于美国获准用于医疗用途。[11]此药物已列入世界卫生组织基本药物标准清单中。[12][13]它有通用名药物,且以许多品牌在全球销售。[1]它是美国于2020年排名第162最常使用的处方药,开立的处方笺数量超过300万张。[14][15]
医疗用途

此药物主要用于治疗单纯疱疹病毒、水痘和带状疱疹感染,包括:[2][16][17]
- 生殖器单纯疱疹病毒(治疗和预防)
- 新生儿单纯疱疹病毒感染
- 唇疮
- 带状疱疹
- 免疫缺陷者的急性水痘感染
- 单纯疱疹病毒脑炎
- 免疫缺陷者的急性皮肤黏膜交界处单纯疱疹病毒
- 单纯疱疹性角膜炎和单纯疱疹病毒睑缘炎
- 为免疫能力低下族群(如接受癌症化学治疗者)提供疱疹病毒预防[18]
其治疗EBV病毒感染的效力尚不清楚。[6]尚未发现它对治疗EBV引起的传染性单核细胞增多症有效。[19]伐昔洛韦和阿昔洛韦透过抑制病毒的DNA复制而发挥作用,但截至2016年,几乎没有证据显示其对治疗EBV病毒有效。此药物价格昂贵,有引起抗病毒药物抗药性的风险,并且(在1%至10%的病例中)可能会导致患者发生不舒服的副作用。[20]
口服形式的阿昔洛韦似乎不会将罹患带状疱疹个体的疼痛风险降低。[21]使用阿昔洛韦治疗眼部疱疹患者,可能比使用碘苷更为有效及安全。[22]目前尚不清楚含阿昔洛韦眼药水是否比含溴夫定眼药水更为有效。[22]
静脉注射形式的阿昔洛韦可有效治疗不同种类疱疹病毒引起的严重状况,包括严重局部感染、严重生殖器感染、水痘和疱疹病毒脑炎。它对全身性或创伤性疱疹感染、疱疹性湿疹和单纯疱疹病毒脑炎的治疗也有效。对20世纪80年代发表的研究报告审查,显示如果于疫情爆发初期使用阿昔洛韦,可在减少发病数量和持续时间方面发挥一定作用。[23]研究显示外用阿昔洛韦在疫情爆发初期和晚期使用均有效,这项审查采用更严格的方法和更大的样本量,因此具有更高的可信度。[24]在试验中显示药物对预防HIV传播并无作用,但可帮助未接受抗反转录病毒药物(ART)治疗的人群减缓HIV疾病的进展。这项发现强调对HIV感染者中测试简单、廉价的非ART处理策略(例如使用阿昔洛韦和复方新诺明)的重要性。[25]
阿昔洛韦被美国食品药物管理局(FDA)归类为B类药物(药物在动物研究中并未显示出对胎儿的风险,但尚无足够的怀孕妇女研究来确定其安全性),[26]美国疾病管制与预防中心(CDC)和其他机构已于2005年宣布在生殖器疱疹严重复发或首次发作期间,可使用阿昔洛韦作治疗。[27]对于严重的HSV感染(尤其是传播性HSV),也可使用静脉注射形式进行治疗。[28]
对小鼠、兔子和大鼠(使用超过相当于人类使用剂量的10倍以上)在器官形成过程中所做的研究未证明会导致出生缺陷。[29]在对大鼠进行的研究中,于怀孕第10天给予相当于人类标准稳态浓度63倍的药物,[Note 1]会出现头部和尾部发生异常的结果。[29]
CDC于2006年推荐使用阿昔洛韦于治疗个体于怀孕期间的水痘感染,特别是在中期和晚期。[30]
阿昔洛韦会在乳汁中出现,因此建议进行母乳哺育的妇女应予谨慎。有限的试验研究显示母亲口服阿昔洛韦后,接受哺乳的婴儿每天会接触到大约0.3毫克/公斤的剂量。若哺乳期母亲乳房附近或乳房有疱疹病症,应避免亲自哺乳。[26][31]
不良影响
使用阿昔洛韦作全身性治疗(口服或静脉注射)常见的药物不良反应(≥1%的患者)包括恶心、呕吐、腹泻、脑病变(仅发生于静脉注射形式)、注射部位反应和头痛。有报导在高剂量使用时会出现幻觉。不常见的不良反应(0.1-1%的患者)有烦躁、眩晕、困惑、头晕、水肿、关节痛、喉咙痛、便秘、腹痛、脱发、皮疹和虚弱。罕见的不良反应(<0.1%的患者)有昏迷、癫痫发作、嗜中性白血球低下、白血球减少症、尿结晶、厌食、疲倦、肝炎、史蒂芬斯-强森症候群、中毒性表皮坏死松解症、血栓性血小板减少性紫癜、过敏性休克[16]和科塔尔症候群。
由于静脉注射阿昔洛韦会造成肾脏中尿结晶,会对5%至10%的使用者造成可逆性肾毒性。对于脱水和已存在肾功能不全的患者进行快速输注阿昔洛韦有较高尿结晶肾病变风险。充足摄取水分、减缓输注速度以及根据患者肾功能而调整剂量将可将这类风险降低。[32][33][34]
阿昔洛韦代谢产物9-羧基甲氧基甲基鸟嘌呤(9-CMMG) 已被证明会产生神经系统不良事件,特别是对老年人和肾功能不全的人群。[35][36][37]
含阿昔洛韦的外用乳霜通常会导致(≥1%) 的个体皮肤干燥、剥落或短暂刺痛/灼热感。罕见的不良反应有红斑或发痒。[16]当施用于眼睛时,通常会导致(≥1%)的个体发生短暂的轻微刺痛。在眼部使用阿昔洛韦,有少数(0.1-1%的患者)会发生角膜点状上皮擦伤或是过敏反应。[16]
药物交互作用
在体外进行复制研究发现酮康唑与阿昔洛韦并用时,对HSV-1和HSV-2的感染具有协同、剂量相关性抗病毒活性。然而这种效应尚未在临床试验上得到证实,因此需进行更多研究来评估协同作用的实际潜力。[38]
针对丙磺舒与阿昔洛韦同时给药的研究显示阿昔洛韦的生物半衰期可延长,受到尿液和肾清除的速率被降低。[26]
干扰素与阿昔洛韦合并使用时会产生协同作用,对接受静脉注射干扰素的患者于施用阿昔洛韦时应谨慎。[39]
虽然HIV感染者经常会将齐多夫定与阿昔洛韦一起使用,但至少一名患者在接受静脉注射阿昔洛韦30-60天后出现极度嗜睡和昏睡的神经毒性,停用阿昔洛韦后症状得到舒缓。[40]
体液中检测
可对血浆或血清中的阿昔洛韦进行数量检测,以确认肾功能不全患者的药物蓄积,或是诊断急性用药过量中毒状况。[41]
作用机制


阿昔洛韦是一种模仿鸟苷的核苷类似物,其作用是抑制病毒生产DNA的能力。
阿昔洛韦由病毒的胸苷激酶转化为阿昔洛韦单磷酸,然后由宿主细胞激酶转化为阿昔洛韦三磷酸(ACV-TP,也称为阿昔洛-GTP)[29]。ACV-TP是种非常有效的病毒DNA复制抑制剂。 ACV-TP会竞争性抑制及灭活病毒DNA聚合酶[42]。其单磷酸盐也会并入病毒DNA之中,导致蛋白质合成链终止[43][44][29][45][46]。
免疫系统健康的人发生阿昔洛韦抗药性的案例很少见,但在长期接受抗病毒药预防的免疫缺陷人群(移植接受者、因HIV感染而患有后天免疫缺陷症候群者)中较为常见(高达10%)。 对HSV的抗药性机制有病毒胸苷激酶缺陷,以及病毒胸苷激酶或DNA聚合酶的突变,而改变受质敏感性。[47][48]

药物动力学
阿昔洛韦的水溶性不佳,口服形式的生物利用度不高(15-30%),因此如果需要高浓度的效果,则需透过静脉注射的形式。口服给药后1-2小时后出现最大血药浓度。根据美国生物制药分类系统(BCS),阿昔洛韦属于BCS III 类药物,即具可溶性但肠道渗透性低。[51]阿昔洛韦分布率高,报导的蛋白质结合率为9%至33%。[52]阿昔洛韦的生物半衰期(t½)取决于年龄:新生儿为4小时、1-12岁儿童为2-3小时,而成人为3小时。[2]
历史
阿昔洛韦被视为是种启动抗病毒药物新时代的先发者,它具有极高的选择性和低细胞毒性。[5]它在1970年代中期被发现后,即开始用作有效治疗疱疹病毒家族中大多数(包括单纯疱疹病毒和水痘带状疱疹病毒)引起感染的有效药物。合成阿昔洛韦的基础是一种由加勒比海绵Cryptotethya crypta分离出的核苷。[53][54][55]它是由Howard Schaeffer与罗伯·文思、S. Bittner和S. Gurwara共同发现的腺苷类似物 - 无环腺苷,具有不错的抗病毒活性。[56]后来Schaeffer加入葛兰素史克,与药理学家 格特鲁德·B·埃利恩继续开发此药物。[57]一项于1979年发布关于阿昔洛韦的美国专利中将Schaeffer列为发明人。[58]罗伯·文思后来发明阿巴卡韦- 一种用于HIV患者的逆转录酶抑制剂。[59]埃利恩则因使用开创性药物设计于新药物开发而荣获1988年诺贝尔生理学或医学奖。
伐昔洛韦是阿昔洛韦的一种前药,于1995年核准用于医疗用途,经摄取后在体内转化为阿昔洛韦。[60]
阿昔洛韦加上氢羟肾上腺皮质素的乳霜复方药于2009年在美国获得核准上市(商品名为Xerese),用于治疗早期复发性唇疮,以减少成人和儿童(6岁以上)发展成疡性唇疮的可能性,并将愈合时间缩短。[61][62]
社会与文化
阿昔洛韦最初以Zovirax品牌上市,专利于20世纪90年代到期,此后均为通用名药物,以许多品牌在全球销售。[1]
附注
参考文献
延伸阅读
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
