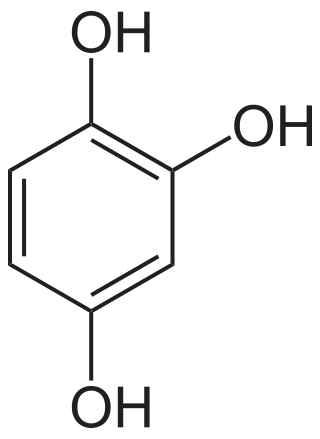
Hydroxyquinol
Chemical compound From Wikipedia, the free encyclopedia
Hydroxyquinol is an organic compound with the formula C6H3(OH)3. It is one of three isomeric benzenetriols. The compound is a colorless solid that is soluble in water. It reacts with air to give a black insoluble solid.[1]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Benzene-1,2,4-triol | |
| Other names
Hydroxyhydroquinone 1,2,4-Benzenetriol 1,2,4-Trihydroxybenzene Benzene-1,2,4-triol 4-Hydroxycatechol 2,4-Dihydroxyphenol 1,3,4-Benzenetriol 1,3,4-Trihydroxybenzene | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.797 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H6O3 | |
| Molar mass | 126.11 g/mol |
| Appearance | white solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Production
It is prepared industrially by acetylation of paraquinone with acetic anhydride followed by hydrolysis of the triacetate.[1]
Historically, hydroxyquinol was produced by the action of potassium hydroxide on hydroquinone.[2] It can also be prepared by dehydrating fructose.[3][4]
- C6H12O6 → 3 H2O + C6H6O3
Natural occurrence
Hydroxyquinol is a common intermediate in the biodegradation of many aromatic compounds. These substrates include monochlorophenols, dichlorophenols, and more complex species such as the pesticide 2,4,5-T.[5] Hydroxyquinol commonly occurs in nature as a biodegradation product of catechin, a natural phenol found in plants (e.g. by soil bacteria Bradyrhizobium japonicum).[6] Hydroxyquinol is also a metabolite in some organisms. For instance, Hydroxyquinol 1,2-dioxygenase is an enzyme that uses hydroxyquinol as a substrate with oxygen to produce 3-hydroxy-cis,cis-muconate.
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.