Thorium dioxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Thorium dioxide?
Summarize this article for a 10 year old
Thorium dioxide (ThO2), also called thorium(IV) oxide, is a crystalline solid, often white or yellow in colour. Also known as thoria, it is mainly a by-product of lanthanide and uranium production.[4] Thorianite is the name of the mineralogical form of thorium dioxide. It is moderately rare and crystallizes in an isometric system. The melting point of thorium oxide is 3300 °C – the highest of all known oxides. Only a few elements (including tungsten and carbon) and a few compounds (including tantalum carbide) have higher melting points.[6] All thorium compounds, including the dioxide, are radioactive because there are no stable isotopes of thorium.
 | |
| Names | |
|---|---|
| IUPAC names
Thorium dioxide Thorium(IV) oxide | |
| Other names
Thoria Thorium anhydride | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.013.842 |
| EC Number |
|
| 141638 | |
PubChem CID |
|
| UNII | |
| UN number | 2910 2909 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| ThO2 | |
| Molar mass | 264.037 g/mol[1] |
| Appearance | white solid[1] |
| Odor | odorless |
| Density | 10.0 g/cm3[1] |
| Melting point | 3,350 °C (6,060 °F; 3,620 K)[1] |
| Boiling point | 4,400 °C (7,950 °F; 4,670 K)[1] |
| insoluble[1] | |
| Solubility | insoluble in alkali slightly soluble in acid[1] |
| −16.0·10−6 cm3/mol[2] | |
Refractive index (nD) |
2.200 (thorianite)[3] |
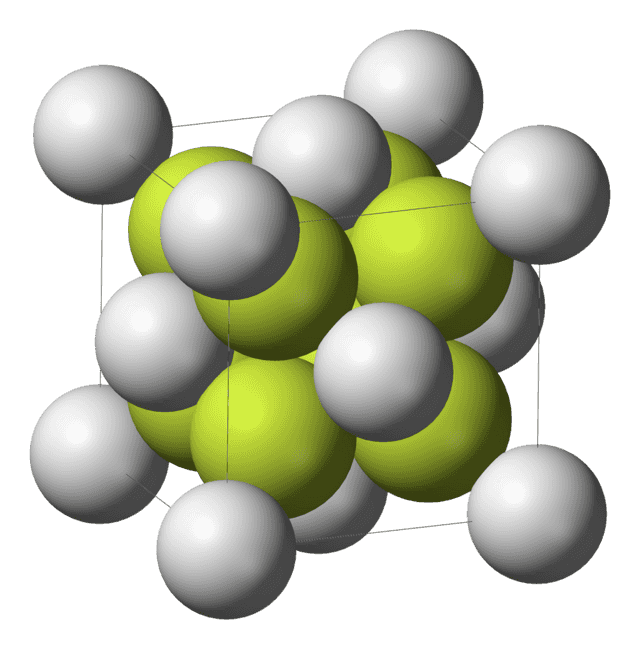
| Structure | |
| Fluorite (cubic), cF12 | |
| Fm3m, No. 225 | |
| Tetrahedral (O2−); cubic (ThIV) | |
| Thermochemistry | |
Std molar entropy (S⦵298) |
65.2(2) J K−1 mol−1 |
Std enthalpy of formation (ΔfH⦵298) |
−1226(4) kJ/mol |
| Hazards | |
| GHS labelling:[5] | |
  | |
| Danger | |
| H301, H311, H331, H350, H373 | |
| P203, P260, P261, P264, P270, P271, P280, P301+P316, P302+P352, P304+P340, P316, P318, P319, P321, P330, P361+P364, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
400 mg/kg |
| Related compounds | |
Other anions |
Thorium(IV) sulfide |
Other cations |
Hafnium(IV) oxide Cerium(IV) oxide |
Related compounds |
Protactinium(IV) oxide Uranium(IV) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
