
Uranium dioxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Uranium(IV) oxide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Uranium dioxide or uranium(IV) oxide (UO2), also known as urania or uranous oxide, is an oxide of uranium, and is a black, radioactive, crystalline powder that naturally occurs in the mineral uraninite. It is used in nuclear fuel rods in nuclear reactors. A mixture of uranium and plutonium dioxides is used as MOX fuel. Prior to 1960, it was used as yellow and black color in ceramic glazes and glass.
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC names
Uranium dioxide Uranium(IV) oxide | |
| Other names
Urania Uranous oxide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.014.273 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| UO2 | |
| Molar mass | 270.03 g/mol |
| Appearance | black powder |
| Density | 10.97 g/cm3 |
| Melting point | 2,865 °C (5,189 °F; 3,138 K) |
| insoluble | |
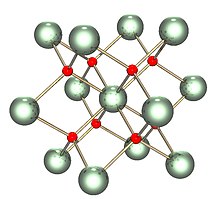
| Structure | |
| Fluorite (cubic), cF12 | |
| Fm3m, No. 225 | |
| Tetrahedral (O2−); cubic (UIV) | |
| Thermochemistry | |
Std molar entropy (S⦵298) |
78 J·mol−1·K−1[2] |
Std enthalpy of formation (ΔfH⦵298) |
−1084 kJ·mol−1[2] |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H300, H330, H373, H410 | |
| P260, P264, P270, P271, P273, P284, P301+P310, P304+P340, P310, P314, P320, P321, P330, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | N/A |
| Safety data sheet (SDS) | ICSC 1251 |
| Related compounds | |
Other anions |
Uranium(IV) sulfide Uranium(IV) selenide |
Other cations |
Protactinium(IV) oxide Neptunium(IV) oxide |
| Triuranium octoxide Uranium trioxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
