
Pethidine
Opioid analgesic / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Pethidine?
Summarize this article for a 10 year old
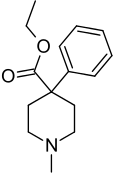
Pethidine, also known as meperidine and sold under the brand name Demerol among others, is a fully synthetic opioid pain medication of the phenylpiperidine class.[5][6][7][8][9][10] Synthesized in 1938[11] as a potential anticholinergic agent by the German chemist Otto Eisleb, its analgesic properties were first recognized by Otto Schaumann while working for IG Farben, in Germany.[12] Pethidine is the prototype of a large family of analgesics including the pethidine 4-phenylpiperidines (piminodine, anileridine and others), the prodines (alphaprodine, MPPP, etc.), bemidones (ketobemidone, etc.) and others more distant, including diphenoxylate and analogues.[13]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Demerol |
| Other names | Meperidine (USAN US) |
| Pregnancy category |
|
| Dependence liability | High |
| Addiction liability | High[1] |
| Routes of administration | By mouth, IV, IM, IT,[2] SC, epidural[3] |
| Drug class | opioid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 50–60% (Oral), 80–90% (Oral, in cases of hepatic impairment) |
| Protein binding | 65–75% |
| Metabolism | Liver: CYP2B6, CYP3A4, CYP2C19, Carboxylesterase 1 |
| Metabolites | • Norpethidine • Pethidinic Acid • others |
| Elimination half-life | 2.5–4 hours, 7–11 hours (liver disease) |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.299 |
| Chemical and physical data | |
| Formula | C15H21NO2 |
| Molar mass | 247.338 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Pethidine is indicated for the treatment of moderate to severe pain, and is delivered as a hydrochloride salt in tablets, as a syrup, or by intramuscular, subcutaneous, or intravenous injection. For much of the 20th century, pethidine was the opioid of choice for many physicians; in 1975, 60% of doctors prescribed it for acute pain and 22% for chronic severe pain.[14]
It was patented in 1937 and approved for medical use in 1943.[15] Compared with morphine, pethidine was considered to be safer, carry a lower risk of addiction, and to be superior in treating the pain associated with biliary spasm or renal colic due to its putative anticholinergic effects.[7] These were later discovered to be inaccurate assumptions, as it carries an equal risk of addiction, possesses no advantageous effects on biliary spasm or renal colic compared to other opioids. Due to the neurotoxicity of its metabolite, norpethidine, it is more toxic than other opioids—especially during long-term use.[7] The norpethidine metabolite was found to have serotonergic effects, so pethidine could, unlike most opioids, increase the risk of triggering serotonin syndrome.[7][8]