
Lindane
Organochlorine chemical and an isomer of hexachlorocyclohexane / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lindane?
Summarize this article for a 10 year old
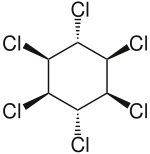
Lindane, also known as gamma-hexachlorocyclohexane (γ-HCH), gammaxene, Gammallin and benzene hexachloride (BHC),[3] is an organochlorine chemical and an isomer of hexachlorocyclohexane that has been used both as an agricultural insecticide and as a pharmaceutical treatment for lice and scabies.[4][5]
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682651 |
| Pregnancy category |
|
| Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 91% |
| Metabolism | Hepatic cytochrome P-450 oxygenase system |
| Elimination half-life | 18 hours |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.365 |
| Chemical and physical data | |
| Formula | C6H6Cl6 |
| Molar mass | 290.81 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Lindane is a neurotoxin that interferes with GABA neurotransmitter function by interacting with the GABAA receptor-chloride channel complex at the picrotoxin binding site. In humans, lindane affects the nervous system, liver, and kidneys, and may well be a carcinogen.[6][7] Whether lindane is an endocrine disruptor is unclear.[8][9][10]
The World Health Organization classifies lindane as "moderately hazardous", and its international trade is restricted and regulated under the Rotterdam Convention on Prior Informed Consent.[11] In 2009, the production and agricultural use of lindane was banned under the Stockholm Convention on persistent organic pollutants.[2][12] A specific exemption to that ban allows it to continue to be used as a second-line pharmaceutical treatment for lice and scabies.[13]