
Hesperidin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Hesperidin?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Hesperidin is a flavanone glycoside found in citrus fruits. Its aglycone is hesperetin. Its name is derived from the word "hesperidium", for fruit produced by citrus trees.
Not to be confused with hesperadin.
Quick Facts Names, Identifiers ...
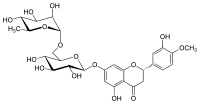 | |
 | |
| Names | |
|---|---|
| IUPAC name
(2S)-3′,5-Dihydroxy-4′-methoxy-7-[α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranosyloxy]flavan-4-one | |
| Systematic IUPAC name
(22S,42S,43R,44S,45S,46R,72R,73R,74R,75R,76S)-13,25,43,44,45,73,74,75-Octahydroxy-14-methoxy-76-methyl-22,23-dihydro-24H-3,6-dioxa-2(2,7)-[1]benzopyrana-4(2,6),7(2)-bis(oxana)-1(1)-benzenaheptaphan-24-one | |
| Other names
Hesperetin, 7-rutinoside,[1] Cirantin, hesperidoside|heperetin, 7-rhamnoglucoside, hesperitin, 7-O-rutinoside | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.007.536 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C28H34O15 | |
| Molar mass | 610.565 g·mol−1 |
| Density | 1.65 ± 0.1g/mL (predicted) |
| Melting point | 262 °C |
| Boiling point | 930.1 ± 65 °C (predicted) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Hesperidin was first isolated in 1828 by French chemist M. Lebreton from the white inner layer of citrus peels (mesocarp, albedo).[2][3]
Hesperidin is believed to play a role in plant defense.