
Cabozantinib
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cabozantinib?
Summarize this article for a 10 year old
Cabozantinib, sold under the brand names Cometriq and Cabometyx among others, is an anti-cancer medication used to treat medullary thyroid cancer, renal cell carcinoma, and hepatocellular carcinoma.[9][10] It is a small molecule inhibitor of the tyrosine kinases c-Met and VEGFR2, and also inhibits AXL and RET.[8][7] It was discovered and developed by Exelixis Inc.
 | |
| Clinical data | |
|---|---|
| Trade names | Cometriq, Cabometyx, others |
| Other names | XL184, BMS907351, cabozantinib s-malate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a613015 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | ≥99.7% |
| Metabolism | Liver (CYP3A4-mediated) |
| Elimination half-life | 110 hours |
| Excretion | Feces (54%), urine (27%) |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.221.147 |
| Chemical and physical data | |
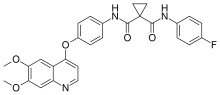
| Formula | C28H24FN3O5 |
| Molar mass | 501.514 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
In November 2012, cabozantinib in its capsule formulation was approved by the US Food and Drug Administration (FDA) under the name Cometriq for treating people with medullary thyroid cancer.[13][14] The capsule form was approved in the European Union for the same purpose in 2014.[11] In April 2016, the FDA granted approval for marketing the tablet formulation (Cabometyx) as a second line treatment for kidney cancer[15][16] and the same was approved in the European Union in September of that year.[12] The brands Cometriq and Cabometyx have different formulations and are not interchangeable.[17]