
Potassium sulfide
Chemical compound / From Wikipedia, the free encyclopedia
Potassium sulfide is an inorganic compound with the formula K2S. The colourless solid is rarely encountered, because it reacts readily with water, a reaction that affords potassium hydrosulfide (KSH) and potassium hydroxide (KOH). Most commonly, the term potassium sulfide refers loosely to this mixture, not the anhydrous solid.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Potassium sulfide | |
| Other names
Dipotassium monosulfide, Dipotassium sulfide, Potassium monosulfide, Potassium sulfide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.013.816 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1847 1382 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| K2S | |
| Molar mass | 110.262 g/mol |
| Appearance | pure: colourless impure: yellow-brown |
| Odor | H2S |
| Density | 1.74 g/cm3 |
| Melting point | 840 °C (1,540 °F; 1,110 K) |
| Boiling point | 912 °C (1,674 °F; 1,185 K) (decomposes) |
| converts to KSH, KOH | |
| Solubility in other solvents | soluble in ethanol, glycerol insoluble in ether |
| −60.0·10−6 cm3/mol | |
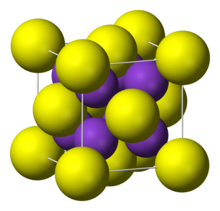
| Structure | |
| antiFluorite | |
| Thermochemistry | |
Std molar entropy (S⦵298) |
105.00 J·mol−1·K−1[1] |
Std enthalpy of formation (ΔfH⦵298) |
-406.2 kJ·mol−1[2] |
Gibbs free energy (ΔfG⦵) |
-392.4 kJ·mol[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Causes skin burns. Dangerous for the environment |
| GHS labelling: | |
  | |
| Danger | |
| H314, H400 | |
| P260, P264, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P391, P405, P501 | |
| Related compounds | |
Other anions |
Potassium oxide Potassium selenide Potassium telluride Potassium polonide |
Other cations |
Lithium sulfide Sodium sulfide Rubidium sulfide Caesium sulfide |
Related compounds |
Potassium hydrosulfide Potassium sulfite Potassium sulfate Iron(II) sulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
