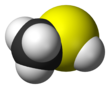
Methanethiol
From Wikipedia, the free encyclopedia
Methanethiol (methanethiol /ˌmɛθ.eɪn.ˈθaɪ.ɒl/, MeSH, (mā kiò-tsò methyl mercaptan); kah-ki liû-sûn (甲基硫醇)) sī tsi̍t-tsióng iú-ki liû huà-ha̍p-bu̍t [en] (有機硫化合物), huà-ha̍k sik sī CH
3SH. Methanethiol sī tsi̍t-tsióng bô-sik ê khì-thé, kū-iú to̍k-ti̍k ê àu-tshàu khì-bī. Methanethiol sī tsi̍t-tsióng thian-jiân ê bu̍t-tsit, tsûn-tsāi teh tōng-bu̍t (pau-kuat jîn-luī) ê hiat-i̍k, tuā-náu hām hùn-piān í-ki̍p si̍t-bu̍t tsoo-tsit tang-tiong. Methanethiol mā thian-jiân tsûn-tsāi teh tsi̍t-kuá-á si̍t-bu̍t tang-tiong, pí-jû tsi̍t-kuá-á kian-kó hām lú-lo̍k (奶酪). Methanethiol sī tì-sú kháu-tshàu hām tn̂g-uī khì (pàng-phuì) tíng-tíng khì-bī ê huà-ha̍p bu̍t tsi-it. Methane-thiol sī siōng kán-tan ê thiol, ū-tang-sî-á sok-siá tsò MeSH. Methanethiol tsiânn-kín to̍h-hué.
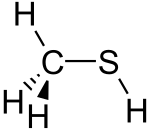 | |||
| |||
| Hō-miâ | |||
|---|---|---|---|
| Iu-sian ê IUPAC hō-miâ
Methanethiol | |||
| Kî-tha hō-miâ
Methyl mercaptan Mercaptomethane Methiol Thiomethyl alcohol/Thiomethanol Methylthiol | |||
| Sek-pia̍t-hō | |||
CAS Number |
| ||
3D model (JSmol) |
|||
| 3DMet | B00105 | ||
| ChEBI | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.000.748 | ||
| EC Number | 200-822-1 | ||
| KEGG |
| ||
PubChem CID |
|||
| RTECS number | PB4375000 | ||
| UNII | |||
| UN number | 1064 | ||
InChI
| |||
SMILES
| |||
| Sèng-chit | |||
| CH3SH | |||
| Mole chit-liōng | 48.11 g·mol−1 | ||
| Gōa-māu | colorless gas[1] | ||
| Khì-bī | Distinctive, like that of rotten cabbage or eggs | ||
| Bi̍t-tō͘ | 0.9 g/mL (liquid at 0°C)[1] | ||
| Iûⁿ-tiám | −123 °C (−189 °F; 150 K) | ||
| Hut-tiám | 5.95 °C (42.71 °F; 279.10 K) | ||
Tī chúi ê iûⁿ-kái-tō͘ |
2% | ||
| Iûⁿ-kái-tō͘ | alcohol, ether | ||
| Cheng-khì-ap | 1.7 atm (20°C)[1] | ||
| Acidity (pKa) | ~10.4 | ||
| Gûi-hiám | |||
| GHS pictograms |     | ||
| GHS signal word | Gûi-hiám | ||
| NFPA 704 | 
4
4
1 | ||
| Ín-hóe-tiám | −18 °C; 0 °F; 255 K[1] | ||
Autoignition temperature |
364 °C; 687 °F; 637 K[2] | ||
| Explosive limits | 3.9%-21.8%[1] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
60.67 mg/kg (tsia̍h-ling tōng-bu̍t)[3] | ||
LC50 (median concentration) |
3.3 ppm (mouse, 2 hr) 675 ppm (rat, 4 hr)[3] | ||
| Bí-kok kiān-hong pī-pha̍k chè-hān (NIOSH): | |||
PEL (Ē-thong-kòe) |
C 10 ppm (20 mg/m3)[1] | ||
REL (Chhui-chiàn) |
C 0.5 ppm (1 mg/m3) [15-minute][1] | ||
IDLH (Chek-sî gûi-hiám) |
150 ppm[1] | ||
| Koan-liân hòa-ha̍p-bu̍t | |||
Koan-liân hòa-ha̍p-bu̍t |
Ethanethiol
Hydrogen sulfide | ||
Tû-liáu te̍k-pia̍t chí chhut, chu-liāu sī kun-kù bu̍t-chit ê piau-chún chōng-thài (tī 25 °C [77 °F], 100 kPa). | |||
| Infobox chham-chiàu | |||