
Citric acid
From Wikipedia, the free encyclopedia
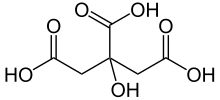
Citric acid is a weak organic acid wi the formula C6H8O7. It is a naitural preservative/conservative an is an aa uised tae add an acidic or sour taste tae fuids an drinks. In biochemistry, the conjugate base o citric acid, citrate, is important as an intermediate in the citric acid cycle, which occurs in the metabolism o aw aerobic organisms.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-hydroxypropane-1,2,3-tricarboxylic acid | |
| Ither names
3-carboxy-3-hydroxypentanedioic acid
2-hydroxy-1,2,3-propanetricarboxylic acid[1] | |
| Identifiers | |
CAS Nummer |
|
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
| EC Nummer | 201-069-1 |
| KEGG |
|
PubChem CID |
|
| RTECS nummer | GE7350000 |
| UNII | |
| |
SMILES
| |
| Properties | |
| C6H8O7 | |
| Molar mass | 192.124 g/mol (anhydrous) 210.14 g/mol (monohydrate) |
| Appearance | crystalline white solid |
| Odour | odorless |
| Density | 1.665 g/cm3(1.5g/cm3 for monohydrate) |
| Meltin pynt | 156 °C (313 °F; 429 K) |
| Bylin pynt | 310 °C (590 °F; 583 K) |
Solubility in watter |
59[2] g/100 ml (20 °C) |
| Solubility | very soluble in ethanol soluble in ether, ethyl acetate insoluble in benzene, chloroform |
| log P | -1.64 |
| Acidity (pKa) | pKa1 = 3.14 pKa2 = 4.75 pKa3 = 6.39,[3] 6.40[4] |
| Thermochemistry | |
| Std enthalpy o formation ΔfH |
-1548.8 kJ/mol |
| Hazards | |
| Main hazards | skin an eye irritant |
| NFPA 704 | 
1
2
0 |
| Flash pynt | 155 °C (311 °F) |
| Explosive leemits | 1.8-4.8% |
| Lethal dose or concentration (LD, LC): | |
LD50 (Median dose) |
3000 mg/kg (rat, oral) |
| Relatit compoonds | |
Except whaur itherwise notit, data are gien for materials in thair staundart state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Close
Citric acid is a commodity chemical, an mair nor a million tonnes are produced every year bi fermentation. It is uised mainly as an acidifier, as a flavorin, an as a chelatin agent.