File:Translational_motion_gif.ogv
Da Wikipedia, l'enciclopedia encyclopedia
Translational_motion_gif.ogv (File video Ogg Theora, durata 19 s, 300 × 264 pixel, 212 kbps, dimensione file: 481 KB)
| Questo file e la sua pagina di descrizione (discussione · modifica) si trovano su Wikimedia Commons (?) |
Dettagli
| DescrizioneTranslational motion gif.ogv |
Motion of gas molecules Español: Animación mostrando la agitación térmica de un gas. Cinco partículas han sido coloreadas de rojo para facilitar el seguimiento de sus movimientos.
Русский: Хаотическое тепловое движение на плоскости частиц газа таких как атомы и молекулы |
| Data | (original) 2010-04-09 (converted to OGV) |
| Fonte |
Questo file deriva da: Translational motion.gif English: Converted as follows:
|
| Autore | A.Greg, en:User:Greg L |
| Altre versioni |
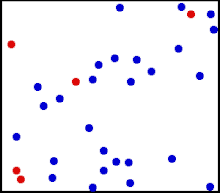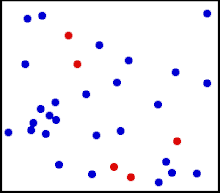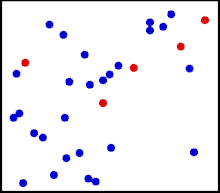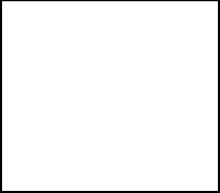 |
Translational motions—the randomized thermal vibrations of fundamental particles such as atoms and molecules—gives a substance its “kinetic temperature.” Here, the size of helium atoms relative to their spacing is shown to scale under 1950 atmospheres of pressure. These room-temperature atoms have a certain, average speed (slowed down here two trillion fold). At any given instant however, a particular helium atom may be moving much faster than average while another may be nearly motionless. The rebound kinetics of elastic collisions are accurately modeled here. If the velocities over time are plotted on a histogram, a Maxwell-Boltzmann distribution curve will be generated. Five atoms are colored red to facilitate following their motions.
Note that whereas the relative size, spacing, and scaled velocity of the atoms shown here accurately represent room-temperature helium atoms at a pressure of 1950 atmospheres, this is a two-dimensional scientific model; the atoms of gases in the real world aren’t constrained to moving in two dimensions in windows precisely one atom thick. If reality worked like this animation, there would be zero pressure on the two faces of the box bounding the Z-axis. The value of 1950 atmospheres is that which would be achieved if room-temperature helium atoms had the same inter-atomic separation in 3-D as they have in this 2-D animation. p=nm¯c2/3V , where ¯c2 is the mean square speed of the molecules. As according to the gas laws for one mole of gas: pV=RT, where T is the thermodynamic temperature, and R is the molar gas constant, it follows that: RT=nm¯c2/3 Thus, the thermodynamic temperature of a gas is proportional to the mean square speed of its molecules. As the average kinetic energy of translation of the molecules is m¯c2/2, the temperature is given by: T=(m¯c2/2)(2n/3R)
Licenza
| Public domainPublic domainfalsefalse |
| Questa semplice figura geometrica non è tutelabile dalle leggi sul diritto d’autore ed è, quindi, di pubblico dominio in quanto costituita da elementi del patrimonio comune. |  |
Didascalie
Elementi ritratti in questo file
raffigura
14 ago 1995
Cronologia del file
Fare clic su un gruppo data/ora per vedere il file come si presentava nel momento indicato.
| Data/Ora | Miniatura | Dimensioni | Utente | Commento | |
|---|---|---|---|---|---|
| attuale | 10:40, 9 apr 2010 | 19 s, 300 × 264 (481 KB) | 84user | == {{int:filedesc}} == {{Information |Description=Motion of gas molecules {{es|Animación mostrando la agitación térmica de un gas. Cinco partículas han sido coloreadas de rojo para facilitar el seguimiento de sus movimientos.}} {{ru|Хаотическ |
Pagine che usano questo file
Nessuna pagina utilizza questo file.
Utilizzo globale del file
Anche i seguenti wiki usano questo file:
- Usato nelle seguenti pagine di ru.wikipedia.org:
Metadati
Questo file contiene informazioni aggiuntive, probabilmente aggiunte dalla fotocamera o dallo scanner usati per crearlo o digitalizzarlo. Se il file è stato modificato, alcuni dettagli potrebbero non corrispondere alla realtà.
| Software |
|
|---|
