
Sitagliptin
Diabetes medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about sitagliptin?
Summarize this article for a 10 year old
Sitagliptin, sold under the brand name Januvia among others, is an anti-diabetic medication used to treat type 2 diabetes.[7] In the United Kingdom it is listed as less preferred than metformin or a sulfonylurea.[8] It is taken by mouth.[7] It is also available in the fixed-dose combination medication sitagliptin/metformin (Janumet, Janumet XR).[7]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /sɪtəˈɡlɪptɪn/ ⓘ |
| Trade names | Januvia, Zituvio, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a606023 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 87% |
| Protein binding | 38% |
| Metabolism | Liver (CYP3A4- and CYP2C8-mediated) |
| Elimination half-life | 8 to 14 h[6] |
| Excretion | Kidney (80%)[6] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.217.948 |
| Chemical and physical data | |
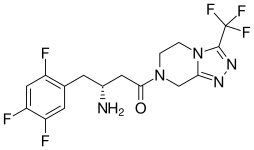
| Formula | C16H15F6N5O |
| Molar mass | 407.320 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Common side effects include headaches, swelling of the legs, and upper respiratory tract infections.[7] Serious side effects may include angioedema, low blood sugar, kidney problems, pancreatitis, and joint pain.[7] Whether use in pregnancy or breastfeeding is safe is unclear.[9] It is in the dipeptidyl peptidase-4 (DPP-4) inhibitor class and works by increasing the production of insulin and decreasing the production of glucagon by the pancreas.[7]
Sitagliptin was developed by Merck & Co. and approved for medical use in the United States in 2006.[7] In 2021, it was the 83rd most commonly prescribed medication in the United States, with more than 8 million prescriptions.[10][11] It is available as a generic medication in Canada but not the United States.[12][13]