Top Qs
Timeline
Chat
Perspective
Valence isomer
Molecule interconvertable with another through pericyclic reactions From Wikipedia, the free encyclopedia
Remove ads
In organic chemistry, two molecules are valence isomers when they are constitutional isomers that can interconvert through pericyclic reactions.[1][2]
Benzene
There are many valence isomers one can draw for the C6H6 formula benzene. Some were originally proposed for benzene itself before the actual structure of benzene was known. Others were later synthesized in lab. Some have been observed to isomerize to benzene, whereas others tend to undergo other reactions instead, or isomerize by ways other than pericyclic reactions.
- Some known valence isomers of benzene
Remove ads
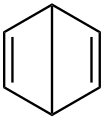
Cyclooctatetraene
Summarize
Perspective
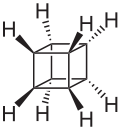
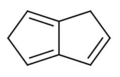
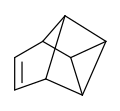
The valence isomers are not restricted to isomers of benzene. Valence isomers are also seen in the series (CH)8. Due to the larger number of units, the number of possible valence isomers is also greater and at least 21:
- Valence isomers of cyclooctatetraene
- Cyclooctatetraene (COT)
- 1,5-dihydropentalene
- 2a,2b,4a,4b-Tetrahydrocyclopropa[cd]pentalene
- Tricyclo[4,2,0,02,5]octa-3,7-diene. The dimer of cyclobutadiene occurs as a cis isomer and a trans isomer. Both isomers convert to COT (symmetry forbidden hence stable) with a half-life of 20 minutes at 140 °C [10]
- Tetracyclo[3,3,0,02,4,03,6] octa-7-ene is only known as its 4-carbomethoxy derivative.[11]
- Tetracyclo[4,2,0,02,4,03,5] octa-7-ene has been prepared from benzvalene and isomerises to COT [12]
- Pentacyclo[5.1.0.02,4. 03,5.06,8]octane (octabisvalene) is the third saturated valence isomer. The (Z)-3,7-phenylsulfonyl derivative is stable up to 200 °C.[13]
Remove ads
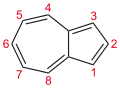
Naphthalene and azulene
Perhaps no pair of valence isomers differ more strongly in appearance than colourless naphthalene and the intensely violet azulene.
- The valence isomers of naphthalene
Benzene oxide and oxepin

References
External links
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads