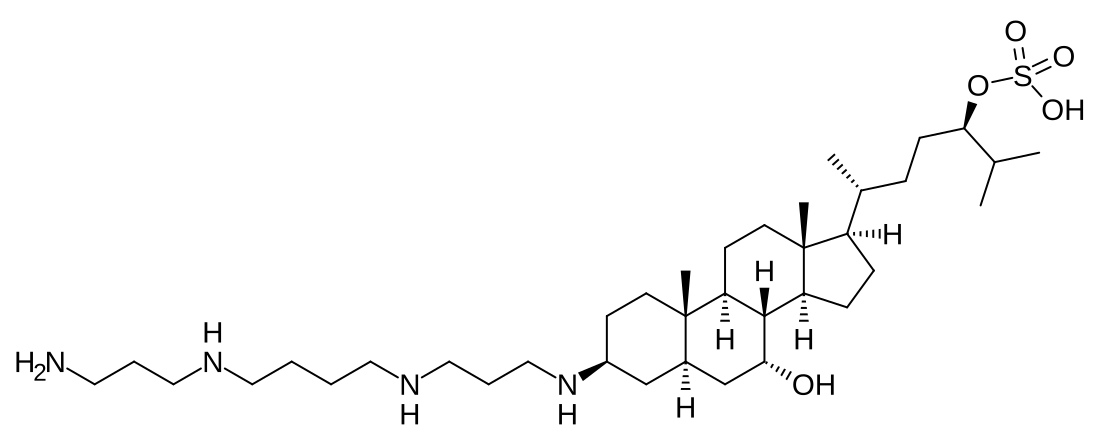
Trodusquemine
Chemical compound From Wikipedia, the free encyclopedia
Trodusquemine is an aminosterol (polyamine steroid conjugate) that inhibits protein tyrosine phosphatase 1B (PTP1B) activity.[1] The compound exhibits broad-spectrum antimicrobial activity[2] and numerous regenerative, neuroprotective, anti-atherosclerotic, antitumor, antiangiogenic, antiobesity, and anxiolytic properties.[3] Phase I clinical trials of trodusquemine have demonstrated good tolerability, but several planned phase II trials were halted due to financial difficulties of the developer.[4]
 | |
| Clinical data | |
|---|---|
| Other names | MSI-1436 |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C37H72N4O5S |
| Molar mass | 685.07 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Chemistry
Trodusquemine is a spermine metabolite of cholesterol. The steroid ring consists of a cholestane with a hydroxyl group at C-7 and sulfate group at C-24; spermine is conjugated to the steroid moiety at C-3. It is structurally similar to squalamine, which features a spermidine moiety instead of spermine.[3]
Pharmacology
Summarize
Perspective
Trodusquemine is a non-competitive allosteric inhibitor of protein tyrosine phosphatase 1B (PTP1B) with an IC50 value of 1 μmol/L.[5] Inhibition of PTP1B prevents dephosphorylation of the insulin receptor, thereby increasing insulin signaling and lowering blood glucose.[4] Trodusquemine also demonstrates affinity for the dopamine transporter (IC50 0.4 μmol/L) and norepinephrine transporter (IC50 0.7 μmol/L).[5]
Trodusquemine suppresses appetite, promotes weight loss, and rescues hyperglycemia in genetic mouse models of obesity (ob/ob) and diabetes (db/db).[6] Other effects of trodusquemine include amelioration of the metabolic syndrome in mouse models of insulin resistance;[7] correction of hepatic steatosis in ob/ob mice;[8] reversal of atherosclerosis in LDLR knock-out mice;[9] inhibition of the growth of malignancy in rodents;[10] stimulation of the regeneration of tail-fin and heart muscle in zebrafish;[11] stimulation of regenerative repair of myocardial infarction and traumatic limb muscle injury in adult mice;[11] prevention of aortic valve calcification in a mouse atheroma model;[12] stimulation of T-cell anti-tumor immunity in a mouse model;[10] correction of systemic and hepatic inflammation, insulin resistance and hepatic dysfunction in horses suffering from equine metabolic syndrome.[13]
Demonstrations of trodusquemine's neuroprotective effects include reversal of memory impairment, normalization of behavior, reduction of neuronal loss and increase in healthspan and lifespan in mouse models of Alzheimer's disease;[14] reduction in alpha-synuclein aggregation and increase in healthspan and lifespan in a C.elegans model of Parkinson's disease;[15] Trodusquemine may exert its effects by targeting specific centers in the brain.[7] Trodusquemine may also have anxiolytic properties.[16]
Although the physiological basis for the healthy lifespan of certain shark species remains unknown, trodusquemine targets well-recognized aging associated processes at both the cellular level and in vivo across many species. These observations conducted in different laboratories suggest that Trodusquemine represents a novel endogenous vertebrate geroprotector.[3]
History
Trodusquemine was originally isolated from liver extracts of the spiny dogfish (Squalus acanthias).[2] It was discovered through a search for antimicrobial compounds in Squaliformes, which lack a robust adaptive immune system. It was hypothesized that their innate immunity might be conferred by endogenous production of antimicrobial compounds.[3]
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.