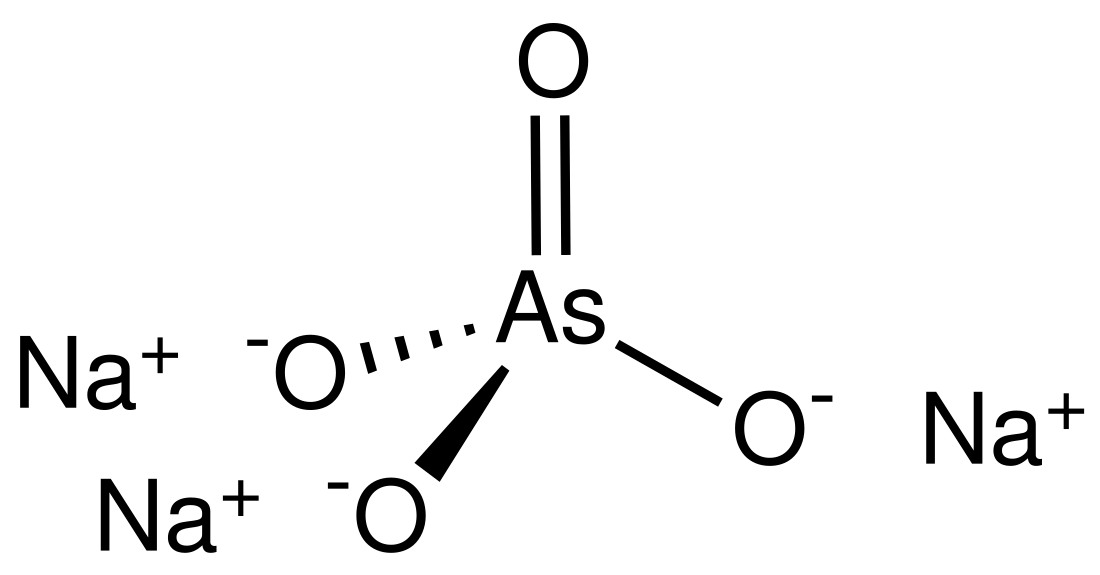
Sodium arsenate
Chemical compound From Wikipedia, the free encyclopedia
Sodium arsenate is the inorganic compound with the formula Na3AsO4. Related salts are also called sodium arsenate, including Na2HAsO4 (disodium hydrogen arsenate) and NaH2AsO4 (sodium dihydrogen arsenate). The trisodium salt is a white or colourless solid that is highly toxic. It is usually handled as the dodecahydrate Na3AsO4.12H2O.[2]
 | |
| Names | |
|---|---|
| IUPAC name
Trisodium arsorate[1] | |
| Other names
sodium arsenate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider | |
| ECHA InfoCard | 100.033.334 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| H24Na3AsO16 (dodecahydrate) | |
| Molar mass | 207.88851 g/mol |
| Appearance | colourless solid |
| Density | 1.517 g/cm3 (dodecahydrate) |
| soluble | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
toxic |
| GHS labelling: | |
   | |
| Danger | |
| H301, H331, H350, H410 | |
| P201, P202, P261, P264, P270, P271, P273, P281, P301+P310, P304+P340, P308+P313, P311, P321, P330, P391, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The compound can be obtained by neutralizing arsenic acid:
- H3AsO4 + 3 NaOH → Na3AsO4 + 3 H2O
The salt (as its dodecahydrate) is isomorphous with trisodium phosphate.[3] The anion AsO43- exists at high pH, but below pH 11.5, it converts to HAsO42- (also written HOAsO32-).
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.