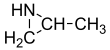Loading AI tools
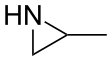
Propyleneimine (or propylene imine) is the organic compound with the formula CH3CH(NH)CH2. It is a secondary amine and the smallest chiral aziridine (ring containing C2N). It is a flammable colorless liquid. Its derivatives, copolymers and oligomers, are of commercial interest.[4]
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Methylaziridine | |||
| Other names
1,2-Propylenimine | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
| ECHA InfoCard | 100.000.799 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1921 (inhibited) | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C3H7N | |||
| Molar mass | 57.096 g·mol−1 | ||
| Appearance | Colorless, oily liquid[1] | ||
| Odor | ammonia-like[1] | ||
| Density | 0.9 g/mL[2] | ||
| Melting point | −63 °C (−81 °F; 210 K)[2] | ||
| Boiling point | 67 °C (153 °F; 340 K)[2] | ||
| Miscible[2] | |||
| Vapor pressure | 112 mmHg (20°C)[1] | ||
| Hazards | |||
| GHS labelling: | |||
     | |||
| Danger | |||
| H225, H300, H310, H318, H330, H350, H411 | |||
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P262, P264, P270, P271, P273, P280, P281, P284, P301+P310, P302+P350, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P320, P321, P322, P330, P361, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |||
| Flash point | −4 °C (25 °F; 269 K)[2] | ||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published) |
500 ppm (rat, 4 hr)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 2 ppm (5 mg/m3) [skin][1] | ||
REL (Recommended) |
Ca TWA 2 ppm (5 mg/m3) [skin][1] | ||
IDLH (Immediate danger) |
Ca [100 ppm][1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
This chemical is used in the paper, textile, rubber and pharmaceutical industries. Propyleneimine is also used in making paint.
The top global producers of this specialty chemical include DuPont, Mitsubishi Chemical Holdings Corporation, Sigma-Aldrich, Dixie Chemical Company, J and K Scientific, Apollo Scientific, Mitsui Chemicals.[5]
The compound is also of interest for the synthesis of dendrimers, a process that exploits the tendency of aziridines to undergo ring-opening reactions.[6] [2]
NIOSH considers propyleneimine a potential occupational carcinogen.
According to a report prepared for the United Church of Christ, propyleneimine emissions from the Dixie Chemical Company facility in Bayport, Pasadena, Texas, are one of the top ten toxicity-weighted air pollutions in the United States.[7]
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.