Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
Potassium chlorite is a potassium salt of chlorous acid (HClO2) having a chemical formula KClO2. It exists as white powder and its annhydrous form easily undergoes decomposition in presence of heat or radiation (especially gamma rays).[1]
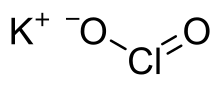 | |
| Names | |
|---|---|
| IUPAC name
potassium;chlorite | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| MeSH | 67063160 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| KClO2, ClKO2 | |
| Molar mass | 106.55 g/mol |
| Hazards | |
| GHS labelling: | |
 | |
| H314 | |
| P260, P264, P280, P301+P330+P331, P304+P340, P305+P351+P338, P310, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium chlorite is a colorless hygroscopic crystal that deliquesces in the air. It decomposes upon heating into potassium chloride and oxygen, emitting light.
Potassium chlorite forms orthorhombic cmcm crystals and has been reported to decompose within hours at room temperature.[1][2] It is an oxidizing agent.
Some of the methods of preparation of potassium chlorite are:
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.