List of cocaine analogues
From Wikipedia, the free encyclopedia
This is a list of cocaine analogues. A cocaine analogue is an (usually) artificial construct of a novel chemical compound from (often the starting point of natural) cocaine's molecular structure, with the result product sufficiently similar to cocaine to display similarity in, but alteration to, its chemical function. Within the scope of analogous compounds created from the structure of cocaine, so named "cocaine analogues" retain 3β-benzoyloxy or similar functionality (the term specifically used usually distinguishes from phenyltropanes, but in the broad sense generally, as a category, includes them) on a tropane skeleton, as compared to other stimulants of the kind. Many of the semi-synthetic cocaine analogues proper which have been made & studied have consisted of among the nine following classes of compounds:[a]
- stereoisomers of cocaine
- 3β-phenyl ring substituted analogues
- 2β-substituted analogues
- N-modified analogues of cocaine
- 3β-carbamoyl analogues
- 3β-alkyl-3-benzyl tropanes
- 6/7-substituted cocaines
- 6-alkyl-3-benzyl tropanes
- piperidine homologues of cocaine
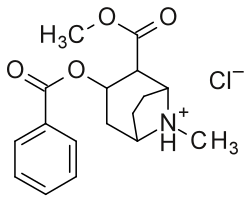
Top: Cocaine in the chair conformation of the tropane-ring, with only its tropane locants given.
Middle: Cocaine with its numerical substitution position locants.
2′ (6′) = ortho, 3′ (5′) = meta & 4′ = para
Bottom: Alternate two-dimensional molecular diagram of cocaine; shown specifically as a protonated, NH+, hydrochloride, and disregarding 3D stereochemistry
Middle: Cocaine with its numerical substitution position locants.
2′ (6′) = ortho, 3′ (5′) = meta & 4′ = para
Bottom: Alternate two-dimensional molecular diagram of cocaine; shown specifically as a protonated, NH+, hydrochloride, and disregarding 3D stereochemistry
However strict analogues of cocaine would also include such other potential combinations as phenacyltropanes & other carbon branched replacements not listed above. The term may also be loosely used to refer to drugs manufactured from cocaine or having their basis as a total synthesis of cocaine, but modified to alter their effect & QSAR. These include both intracellular sodium channel blocker anaesthetics and stimulant dopamine reuptake inhibitor ligands (such as certain, namely tropane-bridged-excised, piperidines). Additionally, researchers have supported combinatorial approaches for taking the most promising analogues currently elucidated and mixing them to the end of discovering novel & efficacious compounds to optimize their utilization for differing distinct specified purposes.[b]
Analogs sensu stricto
Summarize
Perspective
Cocaine Stereoisomers
| Structure | Stereoisomer | S. Singh's alphanumeric assignation |
IC50 (nM) [3H]WIN 3542 inhibition to rat striatal membranes Mean error standard ≤5% in all cases |
IUPAC nomenclature |
|---|---|---|---|---|
 |
R-cocaine (Erythroxyline) |
102 | methyl(1R,2R,3S,5S)-3-(benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylate | |
 |
R-pseudococaine (Delcaine, Depsococaine, Dextrocaine, Isococaine, Psicaine.[2]) |
172 | 15800 | methyl(1R,2S,3S,5S)-3-(benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylate |
 |
R-allococaine | 173 | 6160 | methyl(1R,2R,3R,5S)-3-(benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylate |
 |
R-allopseudococaine | 174 | 28500 | methyl(1R,2S,3R,5S)-3-(benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylate |
 |
S-cocaine | 175 | 15800 | methyl(1S,3R,4R,5R)-3-(benzoyl)oxy-8-methyl-8-azabicyclo[3.2.1]octane-4-carboxylate |
 |
S-pseudococaine | 176 | 22500 | methyl(1S,3R,4S,5R)-3-(benzoyl)oxy-8-methyl-8-azabicyclo[3.2.1]octane-4-carboxylate |
 |
S-allococaine | 177 | 9820 | methyl(1S,3S,4R,5R)-3-(benzoyl)oxy-8-methyl-8-azabicyclo[3.2.1]octane-4-carboxylate |
 |
S-allopseudococaine | 178 | 67700 | methyl(1S,3S,4S,5R)-3-(benzoyl)oxy-8-methyl-8-azabicyclo[3.2.1]octane-4-carboxylate |
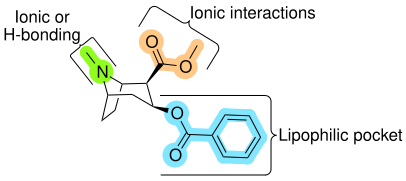
While it was originally thought that the 2β-carbomethoxy moiety interacted with the DAT through hydrogen bonding, subsequent research has indicated that electrostatic (ionic) interactions are the primary means of interactions with the DAT.[c]
There are eight stereoisomers of cocaine (excluding mesomers and modifications to the internal portion of the tropane ring).[d] Due to the presence of four asymmetric carbon atoms in the 1- & 5- to 8 (N) position bond bridge that could adopt R- & S- configurations, cocaine can be considered to have as many as sixteen stereoisomers. However, geometric constraints imparted by the bridgehead amine allow only eight to be created.
The natural isomerism of cocaine is unstable and prone to epimerization. For example, the end product of cocaine biosynthesis contains an axial C2-carbomethoxy moiety which readily undergoes epimerization to the equatorial position via saponification.
For any 2D structural diagrams where stereochemistry is not indicated, it should be assumed the analogue depicted shares the stereochemical conformation of R-cocaine unless noted otherwise.
Arene benzene-ring 2′, 3′, 4′ (5′ & 6′) position (aryl) substitutions
para-substituted benzoylmethylecgonines
| Structure | S. Singh's alphanumeric assignation (name) |
4′=R | DAT
[3H]WIN 35428 |
5-HTT
[3H]Paroxetine |
NET
[3H]Nisoxetine |
Selectivity
5-HTT/DAT |
Selectivity
NET/DAT |
|---|---|---|---|---|---|---|---|
| Cocaine | H | 249 ± 37 | 615 ± 120 | 2500 ± 70 | 2.5 | 10.0 | |
| non-benzoyloxy analogue comparative ligands non-tropane analogue comparative ligands | 11b (WIN 35428) (nisoxetine) (fluoxetine) | F — — | 24 ± 4 775 ± 20 5200 ± 1270 | 690 ± 14 762 ± 90 15 ± 3 | 258 ± 40 135 ± 21 963 ± 158 | 28.7 1.0 0.003 | 10.7 0.2 0.2 |
 | |||||||
| 183a | I | 2522 ± 4 | 1052 ± 23 | 18458 ± 1073 | 0.4 | 7.3 | |
| 183b | Ph | 486 ± 63 | - | - | - | - | |
| 183c | OAc | 144 ± 2 | - | - | - | - | |
| 183d | OH | 158 ± 8 | 3104 ± 148 | 601 ± 11 | 19.6 | 3.8 | |
| (4′-Fluorococaine)[3] | F | - | - | - | - | - | |
| (para-Isothiocyanatobenzoylecgonine methyl ester)[4] (p-Isococ) | NCS | - | - | - | - | - |
| 183a | 183b | 183c |
|---|---|---|
 |  |  |
| 183d | 4'-Fluorococaine | P-ISOCOC |
 |  |  |
The MAT binding pocket analogous to the lipophilic place on cocaine-like compounds, inclusive of the benzene ring, is approximate to 9 Å in length. Which is only slightly larger than a phenyl ring by itself.[f]
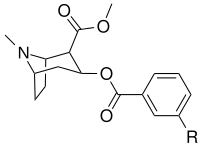
meta-substituted benzoylmethylecgonines
| Structure | S. Singh's alphanumeric assignation (name) |
3′=R | DAT
[3H]WIN 35428 |
5-HTT
[3H]Paroxetine |
NET
[3H]Nisoxetine |
Selectivity
5-HTT/DAT |
Selectivity
NET/DAT |
|---|---|---|---|---|---|---|---|
| (cocaine) | H | 249 ± 37 | 615 ± 120 | 2500 ± 70 | 2.5 | 10.0 | |
 | |||||||
| 184a | I | 325ɑ | - | - | - | - | |
| 184b | OH | 1183 ± 115 | 793 ± 33 | 3760 ± 589 | 0.7 | 3.2 | |
| 191 | OBn | - | - | - | - | - | |
| (m-Isococ) | NCS | - | - | - | - | - |
- ɑIC50 value for displacement of [3H]cocaine
ortho-substituted benzoylmethylecgonines
| Structure | S. Singh's alphanumeric assignation (name) |
2′=R | DAT
[3H]WIN 35428 |
5-HTT
[3H]Paroxetine |
NET
[3H]Nisoxetine |
Selectivity
5-HTT/DAT |
Selectivity
NET/DAT |
|---|---|---|---|---|---|---|---|
| Cocaine | H | 249 ± 37 | 615 ± 120 | 2500 ± 70 | 2.5 | 10.0 | |
 | |||||||
| 185a | I | 350ɑ | - | - | - | - | |
| 185b | F | 604 ± 67 | 1770 ± 309 | 1392 ± 173 | 2.9 | 2.3 | |
| 185c (2′-Acetoxycocaine)[5] | OAc | 70 ± 1 | 219 ± 20 | 72 ± 9 | 3.1 | 1.0 | |
| 185d (2′-Hydroxycocaine)[6] | OH | 25 ± 4 | 143 ± 21 | 48 ± 2 | 5.7 | 1.9 |
- ɑIC50 value for displacement of [3H]cocaine
The hydroxylated 2′-OH analogue exhibited a tenfold increase in potency over cocaine.[i]
Manifold and termination benzoyloxy phenyl-substitutions
Multi-substitutions (substitutions of substitutions; e.g. meta- & para-) or manifold ("many-fold") substituted analogues are analogues where more than one modification from the parent molecule takes place (having numerous intermediary constituents). These are created with often surprising structure–activity relationship results extrapolated therefrom. It is even a common case where two separate substitutions can each yield a weaker, lower affinity or even wholly non-efficacious compound respectively; but due to findings that oftentimes, when used together, such two mutually inferior changes being added in tandem to one analogue has the potential to make the resultant derivative display much greater efficacy, affinity, selectivity &/or strength than even the parent compound; which otherwise was compromised by either of those two alternations when made alone.
| Structure | S. Singh's alphanumeric assignation (name) |
ortho-2′=R | meta-3′=R | para-4′=R | DAT
[3H]WIN 35428 |
5-HTT
[3H]Paroxetine |
NET
[3H]Nisoxetine |
Selectivity
5-HTT/DAT |
Selectivity
NET/DAT |
|---|---|---|---|---|---|---|---|---|---|
 | 186 | HO | H | I | 215 ± 19 | 195 ± 10 | 1021 ± 75 | 0.9 | 4.7 |
 | (Vanillylmethylecgonine)[7] | H | OCH3 | OH | - | - | - | - | - |
| Structure | S. Singh's alphanumeric assignation (name) |
C=R | DAT
[3H]Cocaine (IC50) |
|---|---|---|---|
 | 187 | 1-naphthalene | 742 ± 48 |
 | 188 | 2-naphthalene | 327 ± 63 |
Benzoyl and carbomethoxy branch modifications
Spirocyclic benzoyl branch modification that fits criteria as a cocaine analog and a phenyltropane both (tropane 2nd locant ester rendered in given depiction shows, as has been attested, to only having been successfully alpha configured)[8]

A sulfur in place of the oxygen at the benzoyl ester single bond results in a lower electronegativity than that of cocaine.

REC is a cocaine analogue which contains a "reversed" C2 carbomethoxy moiety. In animal studies, REC lacked cocaine-like stimulant effects.
C1-tropane-ring hydrogen—substitutions
| Structure | Trivial name | R (C1 moiety) |
Ki (nM) @ DAT | Ki (nM) @ SERT | Ki (nM) @ NET | σ1 affinity Ki |
σ2 affinity Ki |
IC50 (μM) Na+ inhibition (Vertridine-Stimulated influx of sodium channels in Neocortical neurons)c |
LogP (XLogP3 algorithm, Cheng et al., 2007) |
|---|---|---|---|---|---|---|---|---|---|
| (—)-Cocaine | H | 326 ± 106 | 513 ± 143 | 358 ± 69 | 6.7 ± 0.3 μMd[13] | "significant"[14] | 6.99 ± 2.43 | 2.30 | |
 | (—)-1-methyl-cocaine | Me | 163 ± 23 | 435 ± 77 | 488 ± 101 | "unappreciable" | 1.13 μM | 16.01 ± 1.90 | 2.67 |
 | (—)-1-ethyl-cocaine | Et | 95.1 ± 17.0ɑ | 1,106 ± 112 | 598 ± 179 | — | — | — | 3.20 |
 | (—)-1-n-propyl-cocaine | n-Pr | 871 ± 205ɑ | 2,949 ± 462b | 796 ± 195 | — | — | — | 3.56 |
 | (—)-1-n-pentyl-cocaine | n-C5H11 | 1,272 ± 199b | 1,866 ± 400ɑ | 1,596 ± 21b | — | — | — | 4.64 |
 | (—)-1-phenyl-cocaine | Ph | 32.3 ± 5.7b | 974 ± 308 | 1,980 ± 99b | 524 nM | 198 nM | 0.29 ± 0.07 | 3.77 |
- ɑ, P < 0.05 compared with (—)-cocaine (one-way ANOVA followed by Dunnett's multiple comparisons test)
- b, P < 0.01 compared with (—)-cocaine (one-way ANOVA followed by Dunnett's multiple comparisons test)
- cLidocaine was found to have a value of 39.6 ± 2.4, the weakest of all tested.
- dSame reference gives 25.9 ± 2.4 μM for (+)-cocaine and 13.6 ± 1.3 μM for norcocaine. Comparably it gives 12.7 ± 1.5 μM for the sigmaergic affinity of (+)-amphetamine. Another reference gives 1.7-6.7 μM for (—)-cocaine. All values Ki.[15]
- Using same data-set as above table, the following compounds were found to compare as:
- CFT @ DAT = 39.2 ± 7.1 (n = 5)
- fluoxetine @ SERT = 27.3 ± 9.2 (n = 3)
- desipramine @ NET = 2.74 ± 0.59 (n = 3)
Cocaine analogs substituting the C1-tropane ring position, requiring sulfinimine (N-sulfinyl-imine) chemistry (before the innovation of which were untenable) which bind unlike the typical configuration at DAT (open to out) as cocaine (with its terminal D79-Y156 distance of 6.03 Å), or in the atypical (closed to out) conformation of the benztropines (3.29 Å). Though closer to the open to out: (—)-1-methyl-cocaine = 4.40 Å & (—)-1-phenyl-cocaine = 4.89 Å, and exhibiting preferential interaction with outward facing DAT conformation, they appear to have the lack of behavioral stimulation as-like the closed to out type. Despite having non-stimulant behavior profiles, they still seem to have anti-depressant behavioral profiles.[12]
The C1 phenyl analog is ten times stronger than cocaine as a dopamine reuptake pump ligand, and twenty four times stronger as a local anesthetic (voltage-dependent Na+ channel blocker), whereas the C1 methyl analog is 2.3 times less potent as a local anesthetic.[12]
cf. hydroxytropacocaine for a natural alkaloid (lacking however, the 2-position carbmethoxy) that is a C1 substituent with a hydroxy group.
2β-substitutions
| Structure | S. Singh's alphanumeric assignation (name) |
R | DAT
[3H]WIN 35428 |
5-HTT
[3H]Paroxetine |
NET
[3H]Nisoxetine |
Selectivity
5-HTT/DAT |
Selectivity
NET/DAT |
|---|---|---|---|---|---|---|---|
 | |||||||
| (Cocaine) | Me | 89 ± 4.8 | 1045 ± 89 | 3298 ± 293 | 11.7 | 37.0 | |
| 196a (Cocaethylene) | Et | 195 ± 45 | 5801 ± 493 | 10000 ± 751 | 29.7 | 51.3 | |
| 196b | n-Pr | 196 ± 46 | 4517 ± 430 | 6124 ± 262 | 23.3 | 31.2 | |
| 196c | i-Pr | 219 ± 48 | 25224 ± 1498 | 30384 ± 1685 | 115 | 139 | |
| 196d | Ph | 112 ± 31 | 33666 ± 3330 | 31024 ± 1909 | 300 | 277 | |
| 196e | Bn | 257 ± 14 | 302 ± 23 | 20794 ± 950 | 1.2 | 80.9 | |
| 196f | β-phenethyl | 181 ± 10 | 615 ± 52 | 19944 ± 1026 | 3.4 | 110 | |
| 196g | γ-phenylpropyl | 147 ± 19 | 374 ± 15 | 4893 ± 344 | 2.5 | 33.3 | |
| 196h | cinnamyl | 371 ± 15 | 368 ± 6.3 | 68931 ± 3476 | 1.0 | 186 | |
| 196i | p-NO2-β-phenethyl | 601 ± 28 | - | - | - | - | |
| 196j | p-Cl-β-phenethyl | 271 ± 12 | - | - | - | - | |
| 196k | p-NH2-β-phenethyl | 72 ± 7 | - | - | - | - | |
| 196l | p-NCS-β-phenethyl | 196 ± 14 | - | - | - | - | |
| 196m | p-azido-β-phenethyl | 227 ± 19 | - | - | - | - | |
| 196n | (p-NHCOCH2Br)β-phenethyl | 61 ± 6 | - | - | - | - | |
| 196o | (p-NHCO(CH2)2CO2Et)β-phenethyl | 86 ± 4 | - | - | - | - | |
 | 197a | NH2 | 753 ± 41.3 | 13725 ± 1256 | 3981 ± 229 | 18.2 | 5.3 |
| 197b | -NMe2 | 127 ± 6.36 | 143713 ± 8854 | 7329 ± 158 | 1131 | 57.7 | |
| 197c | -N(OMe)Me | 60 ± 6.4 | 28162 ± 2565 | 3935 ± 266 | 469 | 65.6 | |
| 197d | -NHMe | 2424 ± 118 | 44798 ± 2105 | 4213 ± 206 | 18.5 | 1.7 | |
| 197e (Benzoylecgonine) | -OH | 195000 | - | - | - | - | |
 | 197f | HOCH2- | 561 ± 149 | - | - | - | - |
| 197g (Tropacocaine) | H | 5180 ± 1160 | - | - | - | - |
| 196a (Cocaethylene) | 196b | 196c | 196d | 196e |
|---|---|---|---|---|
 |  |  |  |  |
| 196f | 196g | 196h | 196i | 196j |
 |  |  |  |  |
| 196k | 196l | 196m | 196n | 196o |
 |  |  |  |  |
| 197a | 197b | 197c | 197d | 197e |
 |  |  |  |  |
| 197f | 197g | |||
 |  | |||
Compounds 196e-h possess greater SERT affinity than cocaine, but possess weaker NET/DAT affinities (with the exception of 196g at NET). Compounds 196k, 196n, 196o, and 197c all possess greater DAT affinity than cocaine. Compound 197b (dimethyl amide) displayed a 1,131-fold increased selectivity in affinity over the serotonin transporter, with only slight reductions in potency for the dopamine & norepinephrine transporters.[m] Whereas 197c (Weinreb amide, N-methoxy-N-methyl amide) had a 469× increase at SERT, with greater affinity for DAT than cocaine and an equal NET affinity.[n] 197b was 137×, and 196c 27× less potent at binding to the serotonin transporter, but both had a NET / DAT ratio that made for a better dopaminergic than cocaine.[o] The consideration that large, bulky C2 substituents would alter the spatial conformation of the tropane ring system by distorting the piperidine portion of the system and thus hamper binding[p] appears to be unfounded.[q]
Benzoylecgonine (197e) is the inactive primary metabolite of cocaine generated through hydrolysis of the C2 methyl ester. In vitro binding studies indicate that benzoylecgonine is ~2,200x less potent than cocaine at the dopamine transporter, possibly due to zwitterion formation preventing strong DAT binding. In contrast to in vitro studies, the lack of activity observed in in vivo studies is likely the result of reduced blood–brain barrier penetration than formation of a zwitterion.[r]
Bioisostere 2-position carbmethoxy-ester functional replacements
| Structure | S. Singh's alphanumeric assignation (name) |
R | [3H]Mazindol | [3H]DA | Selectivity
Uptake/Binding |
|---|---|---|---|---|---|
| (Cocaine) | (H) | 580 ± 70 | 570 ± 180 | 1.0 | |
 | |||||
| 198a | H | 520 ± 40 | 260 ± 70 | 0.5 | |
| 198b | CO2Et (5′-carboethoxy-) | 120 ± 10 | 290 ± 40 | 2.4 | |
| 198c | BOC | 2230 ± 220 | 1820 ± 810 | 0.8 | |
| 198d | Ph | 2000 ± 640 | 2920 ± 1620 | 1.5 | |
| 198e | CH=CHCO2Me | 3600 ± 400 | 3590 ± 1180 | 1.0 | |
 | |||||
| 199a | β(or R)CO2Et | 710 ± 150 | 1060 ± 340 | 1.5 | |
| 199b | α(or S)CO2Et | 5830 ± 630 | 8460 ± 620 | 1.4 | |
 | 200 | 880 ± 350 | 400 ± 140 | 0.4 |
Vinylogous 2β-position carbmethoxy-ester functional replacements
| Structure | S. Singh's alphanumeric assignation |
R | [3H]Mazindol | [3H]DA | Selectivity
Uptake/Binding |
|---|---|---|---|---|---|
 | |||||
| Cocaine | 580 ± 70 | 570 ± 180 | 1 | ||
| 201a | H | 1730 ± 550 | 1120 ± 390 | 0.6 | |
| 201b | Cl | 222 ± 49 | 368 ± 190 | 1.6 | |
| 201c | CO2Et | 50 ± 10 | 130 ± 10 | 2.6 | |
| 201d | CH=CHCO2Et | 1220 ± 100 | 870 ± 50 | 0.7 | |
| 201e | PO(OEt)2 | 4850 ± 470 | 5500 ± 70 | 1.1 |
Compounds 201b & 201c were significantly more potent than cocaine while compounds 201a, 201d & 201e were significantly less potent. This finding indicates that the presence of a hydrogen bond acceptor (i.e. carbomethoxy) at the 2β position is not absolutely necessary for the creation of high affinity cocaine analogues.[w]
N-modifications
| Compound | S. Singh's alphanumeric assignation (name) |
N8-R | [3H]Mazindol binding |
[3H]DA uptake |
Selectivity
Uptake/Binding |
|---|---|---|---|---|---|
 | 217 (Cocaine methiodide) | - | 10700 ± 1530ɑ | - | - |
 | (Cocaine) | CH3 | 280 ± 60 102ɑ | 320 ± 10 | 1.1 |
| 218 (Norcocaine) | H | 303 ± 59ɑ | - | - | |
| 219a | Bn | 668 ± 67ɑ | - | - | |
| 219b | Ac | 3370 ± 1080ɑ | - | - | |
| 219c | CH2CH2OH | 700 ± 100 | 1600 ± 200 | 2.3 | |
| 219d | CH2CO2CH3 | 480 ± 40 | 1600 ± 100 | 3.3 | |
| 219e | CH2CO2H | 380 ± 20 | 2100 ± 400 | 5.5 | |
| 220a | SO2CH3 (Ms) | 1290 ± 80 | 1970 ± 70 | 1.5 | |
| 220b | SO2CF3 (Tf) | 330 ± 30 | 760 ± 20 | 2.3 | |
| 220c | SO2NCO | 120 ± 10 | 160 ± 10 | 1.3 | |
| 220d | SO2Ph | 20800 ± 3500 | 61000 | 2.9 | |
| 220e | SO2C6H4-4-NO2 (nosyl) | 5720 ± 1140 | 18800 ± 90 | 3.3 | |
| 220f | SO2C6H4-4-OCH3 | 6820 ± 580 | 16400 ± 1400 | 2.4 | |
| 221a | NO | 99500 ± 12300 | 231700 ± 39500 | 2.3 | |
| 221b | NO2 | 7500 ± 900 | 21200 ± 600 | 2.8 | |
| 221c | NHCOCH3 | >1000000 | >1000000 | - | |
| 221d | NH2 | - | - | - |
- ɑIC50 (nM) for displacement of [3H]WIN 35428
| Norcocaine (218) | 219a | 219b | 219c |
|---|---|---|---|
 |  |  |  |
| 219d | 219e | 220a | 220b |
 |  |  |  |
| 220c | 220d | 220e | 220f |
 |  |  |  |
| 221a | 221b | 221c | 221d |
 |  |  |  |
Tricyclic cocaine analogues
8 to 2 tethered analogues
| Compound (S. Singh's #) |
Structure | [3H]Mazindol binding | [3H]DA uptake | [3H]5-HT uptake | [3H]NE uptake | selectivity [3H]5-HT/[3H]DA |
|---|---|---|---|---|---|---|
| cocaine | 375 ± 68 | 423 ± 147 | 155 ± 40 | 83.3 ± 1.5 | 0.4 | |
| (–)-128 | 54.3 ± 10.2 | 60.3 ± 0.4 | 1.76 ± 0.23 | 5.24 ± 0.07 | 0.03 | |
| (+)-128 | 79 ± 19 | 114 ± 28 | 1.48 ± 0.07 | 4.62 ± 0.31 | 0.01 | |
| (±)-128 |  | 61.7 ± 8.5 | 60.3 ± 0.4 | 2.32 ± 0.23 | 2.69 ± 0.12 | 0.04 |
| 129 |  | 6.86 ± 0.43 | 24.0 ± 1.3 | 1.77 ± 0.04 | 1.06 ± 0.03 | 0.07 |
| 130a |  | 17.2 ± 1.13 | 10.2 ± 1.4 | 78.9 ± 0.9 | 15.0 ± 0.4 | 7.8 |
| 131a |  | 4.00 ± 0.07 | 2.23 ± 0.12 | 14.0 ± 0.6 | 2.99 ± 0.17 | 6.3 |
| 131b |  | 3.61 ± 0.43 | 11.3 ± 1.1 | 25.7 ± 4.3 | 4.43 ± 0.01 | 2.3 |
| 132a |  | 13.7 ± 0.8 | 14.2 ± 0.1 | 618 ± 87 | 3.84 ± 0.35 | 43.5 |
| 133a |  | 149 ± 6 | 149 ± 2 | 810 ± 80 | 51.7 ± 12 | 5.4 |
See N-front & back bridged phenyltropanes.
| Compound | S. Singh's alphanumeric assignation |
[3H]Mazindol | [3H]DA | Selectivity
Uptake/Binding |
|---|---|---|---|---|
 | 222 | 44900 ± 6200 | 115000 ± 15700 | 2.6 |
Back-bridged cocaine analogues are considered more akin to untethered cocaine analogs & phenyltropane derivatives (where the nitrogen lone pair is not fixed or constrained) and better mimics their affinities. This is due to when the eighth carbon tropane position is freely rotatable and unbound it preferably occupies the axial position as defining its least energy & most unhindered state. In front-bridged analogs the nitrogen lone pairings rigid fixity makes it reside in an equatorial placing for the piperidine ring-part of the tropane nucleus, pointing to the two-carbon & three methylene unit bridgehead; giving the attested front-bridged cocaine analogues preference for SERT over DAT.[aa]
8 to 3 tethered analogues
| Structure | Compound | R | X | [3H]DA Uptake | [3H]5-HT Uptake | [3H]NE Uptake | 5-HT/DA Selectivity | NE/DA Selectivity |
|---|---|---|---|---|---|---|---|---|
| Cocaine | 259 ± 19.9 | 155 ± 0.4 | 108 ± 3.5 | 0.60 | 0.42 | |||
 | ||||||||
| 5a | H | CO2Me | 268 ± 16.6 | 2046 ± 42 | 26.4 ± 1.9 | 7.63 | 0.10 | |
| 5b | Me | CO2Me | 403 ± 20 | 179 ± 38 | 4.9 ± 0.2 | 0.44 | 0.01 | |
| 5c | I | CO2Me | 368 ± 1.6 | 29 ± 1.6 | 5 ± 1.3 | 0.08 | 0.01 | |
| 7 | H | CO2iPr | 428 ± 45.7 | 1150 ± 1.6 | 52.3 ± 12.0 | 2.69 | 0.12 | |
| 8 | H | CH2OH | ~3000 | ~1000 | ~300 | ~0.33 | ~ 0.1 | |
| 9 | H | CH2OAc | 610 ± 53 | 1530 ± 150 | 283 ± 16 | 2.51 | 0.46 | |
| 10 | H | CH2OCOC(CH3)3 | 1020 ± 70 | 168 ± 53.5 | 1180 ± 130 | 0.16 | 1.16 | |
| 11 | H | CH2OCOPh | 1750 ± 140 | 1.53 ± 0.19 | 894 ± 126 | 0.0009 | 0.51 | |
| 12 | H | CH2OCO-2-naphthyl | 1678 ± 124 | 169 ± 16 | 1234 ± 166 | 0.10 | 0.74 | |
| 13 | H | CH2NHCOCH3 | 6140 ± 50 | 13330 ± 3150 | 2430 ± 340 | 2.17 | 0.39 | |
| 14 | H | CH2NHCO2C(CH3)3 | 2300 ± 380 | 2360 ± 30 | 1700 ± 60 | 1.03 | 0.74 |
| Structure | Compound | R | X | [3H]DA Uptake | [3H]5-HT Uptake | [3H]NE Uptake | DA/5-HT Selectivity | NE/DA Selectivity |
|---|---|---|---|---|---|---|---|---|
| Cocaine | 423 ± 147 | 155 ± 0.4 | 108 ± 3.5 | 2.7 | 0.26 | |||
 | ||||||||
| 8a | 4-F | CO2Me | 6620 ± 460 | 335 ± 45 | 584 ± 163 | 2.7 | 0.26 | |
| 8b | 4-Cl | CO2Me | 853 ± 58 | 34.3 ± 2.9 | 208 ± 111 | 24.8 | 0.24 | |
| 8c | 3-Cl | CO2Me | 7780 ± 1580 | 53.6 ± 17.2 | 231 ± 44 | 145 | 0.03 | |
| 8d | 4-Br | CO2Me | 495 ± 13 | 11 ± 3 | 178 ± 9 | 45 | 0.36 | |
| 8e | 4-I | CO2Me | 764 ± 11 | 21.9 ± 0.3 | 213 ± 31 | 34.9 | 0.28 | |
| 8f | 4-CF3 | CO2Me | N/T | 12.6 ± 0.5 | 1830 ± 211 | N/T | N/T | |
| 8g | H | CO2Me | 481 ± 11 | 1140 ± 70 | 53 ± 16 | 0.42 | 0.11 | |
| 8h | 4-Me | CO2Me | 649 ± 2 | 15 ± 0.4 | 146 ± 28 | 43.3 | 0.22 | |
| 8i | 4-OCH3 | CO2Me | 3130 ± 160 | 56 ± 4 | 187 ± 5 | 55.9 | 0.06 | |
| 8j | 4-iPr | CO2Me | N/T | 10.2 ± 0.4 | 1110 ± 200 | N/T | N/T | |
| 8k | 3,4-Cl2 | CO2Me | 1920 ± 260 | 20 ± 1 | 1000 ± 280 | 96 | 0.52 | |
| 8l | 2,3-Cl2 | CO2Me | 950 ± 107 | 354 ± 188 | 1210 ± 358 | 2.4 | 1.42 | |
| 8m | 3,5-Cl2 | CO2Me | 5600 ± 400 | 437 ± 0.3 | 4100 ± 500 | 12.8 | 0.73 | |
| 8n | 3,4-F2 | CO2Me | 7440 ± 19 | 101 ± 8.7 | 394 ± 98 | 73.7 | 0.05 | |
| 8o | 4-Br-3-Cl | CO2Me | 5420 ± 940 | 2.3 ± 0.1 | 459 ± 80 | 2360 | 0.08 | |
| 8p | 3-Cl-4-I | CO2Me | 3140 ± 450 | 1.8 ± 0.3 | 272 ± 55 | 1740 | 0.09 | |
| 8q | 2-Cl-4-I | CO2Me | 6640 ± 2080 | 74 ± 12.2 | 508 ± 21 | 89.7 | 0.08 | |
| 8r | 3-Cl-4-Me | CO2Me | >10000 | 6.4 ± 1.3 | 198 ± 10 | >1560 | <0.02 | |
| 8s | 3,4-Me2 | CO2Me | N/T | 10.1 ± 1.1 | 659 ± 128 | N/T | N/T | |
 | ||||||||
| 8t | 1-Naphthyl | CO2Me | 9720 ± 700 | 121 ± 3 | 5370 ± 580 | 80.3 | 0.55 | |
| 8u | 2-Naphthyl | CO2Me | 735 ± 235 | 21 ± 9.9 | 157 ± 13 | 35 | 0.21 | |
| 8v | 1-Pyrenyl | CO2Me | 9920 ± 906 | 860 ± 20.6 | N/T | 11.5 | N/T | |
| 8w | 9-Phenanthryl | CO2Me | 1640 ± 30 | 233 ± 44 | 13000 ± 1300 | 39.2 | 0.86 |
- "N/T" = "not tested"
Tropane ring contraction (azabornane) analogues

| Structure | S. Singh's alphanumeric assignation (name) |
DAT [3H]WIN 35428 Ki (nM) |
|---|---|---|
| (Cocaine) | 89 ± 4.8 | |
 | 155a | 60400 ± 4800 |
 | 155b | 96500 ± 42 |
 | 155c | 5620 ± 390 |
 | 155d | 18900 ± 1700 |
6/7 tropane position methoxycocaine & methoxypseudococaine analogues

| Compound | S. Singh's alphanumeric assignation (name) |
X | Ki (nM) [3H]Mazindol binding |
Ki (nM) [3H]DA uptake |
Selectivity
Uptake/Binding |
|---|---|---|---|---|---|
| (Cocaine) | 280 ± 60 | 320 ± 10 | 1.1 | ||
| (Pseudococaine) | 10400 ± 300 | 13800 ± 1500 | 1.3 | ||
 | 225a | 2β, 6β-OCH3 | 98000 ± 12000 | 68000 ± 5000 | 0.7 |
 | 225b | 2α, 6β-OCH3 | 190000 ± 11000 | 510000 ± 110000 | 2.7 |
 | 225c | 2β, 7β-OCH3 | 4200 ± 100 | 6100 ± 200 | 1.4 |
 | 225d | 2α, 7β-OCH3 | 45000 ± 5000 | 110000 ± 4000 | 2.4 |
 | 225e | 2α, 7α-OCH3 | 54000 ± 3000 | 200000 ± 70000 | 3.7 |
3β-position 2′—(6′) & 2β-substitution combination analogues
| Compound | S. Singh's alphanumeric assignation |
2β-R | C2′-R | IC50 (nM) (displacement of [3H]WIN 35428) |
|---|---|---|---|---|
 | ||||
| 211a | CH2OH | H | 6214 ± 1269 | |
| 211b | CH2OCOCH3 | H | 2995 ± 223 | |
| 211c | CONHCH3 | H | >100000 | |
| 211d | CO2Et | H | 2031 ± 190 | |
| 211e | CO2-i-Pr | H | 1377 ± 10 | |
| 211f | CO2Ph | H | 2019 ± 253 | |
| 211g | CO2CH2Ph | H | 4602 ± 325 | |
| 211h | 3-phenyl-1,2,4-oxadiazole | H | 3459 ± 60 | |
| 211i | CH=CH2 | H | 2165 ± 253 | |
| 211j | CH2CH3 | H | 2692 ± 486 | |
 | 212 | CO2-i-Pr | HO | 663 ± 70 4507 ± 13ɑ 34838 ± 796b |
- ɑFor displacement of [3H]paroxetine (5-HTT & NET)
- bFor displacement of [3H]nisoxetine (5-HTT & NET)
3β-Carbamoyl analogues
| Compound | S. Singh's alphanumeric assignation (name) |
X | IC50 (nM) inhibition of [3H]Cocaine binding (Rat Striatal Tissue) |
IC50 (nM) inhibition of [3H]DA uptake (Rat Striatal Tissue) |
Selectivity uptake/binding |
|---|---|---|---|---|---|
| (Cocaine) | (H) | 70 ± 10 | 210 ± 70 | 3.0 | |
 | |||||
| 223a | H | 5600 ± 700 | 52600 ± 3000 | 9.4 | |
| 223b | 4-NO2 | 1090 ± 250 | 5700 ± 1200 | 5.2 | |
| 223c | 4-NH2 | 63300 ± 12200 | >100000 | - | |
| 223d | 4-N3 | 1000 ± 240 | 1180 ± 360 | 1.2 | |
| 223e | 4-NCS | 260 ± 60 | 490 ± 80 | 1.9 | |
 | |||||
| 223f | 3-NO2 | 37 ± 10 | 178 ± 23 | 4.8 | |
| 223g | 3-NH2 | 2070 ± 340 | 23100 ± 900 | 11.1 | |
| 223h | 3-N3 | 630 ± 150 | 3900 ± 1590 | 6.2 | |
| 223i | 3-NCS | 960 ± 210 | 4900 ± 420 | 5.1 |
Phenyl 3-position linkage substitutions

The top image above is a 2-Dimensional emulation of the orientation for the animated 3D image to the far right, with a methoxy that is distal from the phenyl group and cis. While the alternate image below that to its bottom shown above is one with the carboxyl methyl group proximal to the phenyl, in its optimum conformation, with a likewise optimum trans configuration.
See: List of phenyltropanes (Many phenyltropanes are derived from cocaine metabolites, such as methylecgonidine, as precursors. Whereas fully synthetic methods have been devised from the starting material of vinylcarbenoids & pyrroles.)[21]
The difference in the length of the benzoyloxy and the phenyl linkage contrasted between cocaine and phenyltropanes makes for a shorter distance between the centroid of the aromatic benzene and the bridge nitrogen of the tropane in the latter PTs. This distance being on a scale of 5.6 Å for phenyltropanes and 7.7 Å for cocaine or analogs with the benzoyloxy intact.[af] This may account for PTs increased behavioral stimulation profile over cocaine.[ag] Differences in binding potency have also been explained considering solvation effects; cocaine containing 2β,3β-ester groups being calculated as more solvated than the WIN-type compounds (i.e. troparil). Higher pKɑs of the tropane nitrogen (8.65 for cocaine, 9.55 for troparil & 11.95 for vinyl analogue 43a), decreased aqueous solvation & decreased conformational flexibility added to increased binding affinity.[ah]
Despite the observation of increased stimulation, phenyltropanes lack the local anesthetic sodium channel blocking effect that the benzoyloxy imparts to cocaine. Beside topical affect, this gives cocaine an affinity for binding to sites on the dopamine and serotonin sodium dependent transport areas that are distinct & specific to MAT in contrast to the general sodium channels; creating a separate mechanism of relational affinity to the transporters in addition to its inhibition of the reuptake for those transporters; this is unique to the local anesthetic value in cocaine & analogues with a similar substitute for the benzoyloxy that leaves the sodium channel blockage ability intact. Rendering such compounds as different functionally in their relation to MAT contrasted to phenyltropane analogues which have the local anesthetic bridge removed.[22] (Requiring some of the sodium ions to be pumped from the axon via Na+/K+-ATPase). In addition, it even has been postulated that a crucial role regarding the electron energy imparted via voltage sensitization (and thus action potential blockage with a molecule capable of intersecting its specific channel, in the case of cocaine a sodium channel, that potentially serves in re-quantifying its charge) upon a receptor binding site may attenuate the mediating influence of the inhibitory regulation that autoreceptors play by their slowing neurotransmitter release when an efflux is created through an instance of agonism by a compound; allowing said efflux to be continued without the body's attempt to maintain homeostasis enacting in as readily responsive a manner to its conformational change.[23]
3β-Alkylphenyltropane & 3β-Alkenyl analogues
| Compound | S. Singh's alphanumeric assignation (name) |
n | IC50 (nM) [3H]Cocaine binding |
IC50 (nM) [3H]DA uptake |
Selectivity uptake/binding |
|---|---|---|---|---|---|
| (Cocaine) | 101 ± 26 | 209 ± 20 | 2.1 | ||
 | |||||
| 224a | 1 | 885 ± 18 | 1020 ± 52 | 1.1 | |
| 224b | 2 | 9.9 ± 0.33 | 70.5 ± 1.0 | 7.1 | |
| 224c | 3 | 344 ± 12 | 2680 ± 190 | 7.8 | |
| 224d | 71.6 ± 0.7 | 138 ± 9 | 1.9 | ||
| 224e | 2.10 ± 0.04 | 5.88 ± 0.09 | 2.8 |
The compound 224e, the 3β-styrene analogue, had the highest potency in its group. While 224b & 224c showed the most selectivity, with 224b having a ten-fold greater potency for the dopamine transporter than cocaine.[aj]
6-Alkyl-3-benzyltropane analogues
| Sub-category (S. Singh compound #) |
a R=H |
b R=Me |
c R=Et |
d R=n-Pr |
e R=n-Bu |
f R=Bn |
|---|---|---|---|---|---|---|
| 2β,6α-isomers: (229a—f) | ||||||
 |  |  |  |  |  | |
| 2α,6α-isomers: (230a—f) | ||||||
 |  |  |  |  |  | |
| 2β,6β-isomers: (231a—f) | ||||||
 |  |  |  |  |  | |
| 2α,6β-isomers: (232a—f) | ||||||
 |  |  |  |  |  | |
| Compound | S. Singh's alphanumeric assignation (name/WIN number) |
R | Ki (nM) [3H]WIN 35428 binding |
IC50 (nM) [3H]DA uptake |
Selectivity
uptake/binding |
|---|---|---|---|---|---|
| Cocaine | 32 ± 5 338 ± 221 | 405 ± 91 405 ± 91 | 12.6 1.2 | ||
| WIN 35065-2 | 33 ± 17 314 ± 222 | 373 ± 10 | 11.3 | ||
 | |||||
| (−)-229a | H | 33 ± 5 | 161 ± 100 | 4.9 | |
| 229a | H | 91 ± 10 | 94 ± 26 | 1.0 | |
| 229b | Me | 211 ± 23 | - | - | |
| 229c | Et | 307 ± 28 | - | - | |
| 229d | n-Pr | 4180 ± 418 | - | - | |
| 229e | n-Bu | 8580 ± 249 | - | - | |
| 229f | Bn | 3080 ± 277 | - | - | |
 | |||||
| (+)-230a | H | 60 ± 6 | 208 ± 63 | 3.5 | |
| 230a | H | 108 ± 14 | 457 ± 104 | 4.2 | |
| 230b | Me | 561 ± 64 | - | - | |
| 230c | Et | 1150 ± 135 | - | - | |
| 230d | n-Pr | 7240 ± 376 | - | - | |
| 230e | n-Bu | 19700 ± 350 | - | - | |
| 230f | Bn | 7590 ± 53 | - | - | |
 | |||||
| 231b | Me | 57 ± 5 | 107 ± 36 | 1.9 | |
| 231c | Et | 3110 ± 187 | - | - | |
| 231d | n-Pr | 5850 ± 702 | - | - | |
| 231f | Bn | 1560 ± 63 | - | - | |
 | |||||
| 232b | Me | 294 ± 29 | 532 ± 136 | 1.8 | |
| 232c | Et | 6210 ± 435 | - | - | |
| 232d | n-Pr | 57300 ± 3440 | - | - | |
| 232f | Bn | 3080 ± 277 | - | - | |
| 241 | Bn | 4830 ± 434 | - | - |
| Sub-category (S. Singh compound #) |
a R=H |
b R=Me |
c R=Et |
d R=n-Pr |
e R=n-Bu |
f R=Bn |
|---|---|---|---|---|---|---|
| 6α-isomers: (237a—f) | ||||||
 |  |  |  |  |  | |
| 6β-isomers (exo): (238a—f) | ||||||
 |  |  |  |  | ||
| 3β-benzyl derivatives: (239a—f) | ||||||
 |  |  |  |  |  | |
| intermediate alkylidene esters: (240a—f) | ||||||
 |  |  |  |  |  | |
N.B. The benzylidene derivatives serve as synthetic intermediates for 6-Alkyl-3-benzyltropanes and have not been assayed for biological activity. Compounds 237a and 238a are the same compound as both are the parent for either series with a hydrogen saturated in their respective substitution place.
Direct 2,3-pyrimidino fused
above: Strobamine, a DARI functional cocaine analog with structural semblance.[24] Compare the phenyltropane length tropane C2 & C3 functional group fusion variant.[25]
below: Chalcostrobamine
below: Chalcostrobamine
cf. strobamine (at right) for a more efficacious compound as like the below.
| Structure | alphanumeric assignation |
R1 | R2 | hDAT IC50 (nM) |
hSERT IC50 (nM) |
hNET IC50 (nM) |
|---|---|---|---|---|---|---|
 | ||||||
| (−)-3a | H | C6H5 | 58,300 (20,200) | 6140 (3350) | NA | |
| (+)-3a | H | C6H5 | 48,700 (20,100) | 6030 (3400) | NA | |
 | ||||||
| (−)-3b | H | NH2 | NA | NA | NA | |
| (+)-3b | H | NH2 | NA | NA | NA | |
 | ||||||
| (−)-3c | H | CH3 | NA | NA | NA | |
| (+)-3c | H | CH3 | NA | NA | NA | |
 | ||||||
| (−)-3d | H | H | NA | NA | NA | |
| (+)-3d | H | H | NA | NA | NA | |
 | (+/—)-3e | C6H5 | C6H5 | 30,000 (11,200) | 3650 (1700) | NA |
- "NA" = "no affinity", e.g. unquantifiable.
Direct di-hetero-benzene (pyrimidino) 2,3-fused and thus rigidified cocaine analogs.[26]
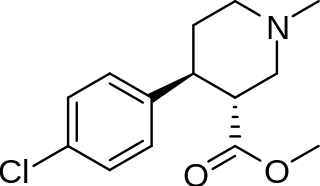
Piperidine cocaine-homologues

| Compound | S. Singh's alphanumeric assignation (name) |
2β-R | IC50 (nM) |
|---|---|---|---|
| (Cocaine) | CO2CH3 (i.e. CO2Me) | 249 ± 37 | |
 | 183a | CO2CH3 | 2522 ± 4 |
 | 242 | H | 11589 ± 4 |
 | 243 | CO2CH3 | 8064 ± 4 |
cf. phenyltropane piperidine-homologues for compounds with a more optimized conformation that yield higher affinities when binding to MAT.
Cocaine hapten analogues

- ɑ6-(2R,3S)-3-(benzoyloxy)-8-methyl-8-azabicyclo [3.2.1] octane-2-carbonyloxy-hexanoic acid
- b6-(2R,3S)-3-(benzoyloxy)-8-methyl-8-azabicyclo [3.2.1] octane-2-carboxamido-hexanoic acid

| Compound | S. Singh's alphanumeric assignation (name) |
R |
|---|---|---|
 | ||
| 401a | CH3 | |
| 401b | (CH2)5CO2H | |
| 401c | CH2CO2H | |
| 401d | COCH2CH2CO2H | |
| 401e | H | |
| 401f | CH2CH2Br | |
| 385g | (CH2)2NHCO(CH2)2CONH2 | |
 | ||
| 402a | O(CH2)4NHCO(CH2)2CO2N(CO2)2C6H4 | |
| 402b | OH | |
| 402c | O(CH2)2(p-NH2C6H4) | |
| 402d | NH(CH2)5CO2H | |
| 402e | O(CH2)4NHCO(CH2)2CONH2 | |
 | ||
| 403a | NH2 | |
| 403b | NHCOCH2Br | |
| 403c | NHCO(CH2)3CO2H | |
| 403d | (CH2)3NHCO(CH2)2CONH2 |
Cocaine haptens that create catalytic anti-bodies require transitional states as affected in vivo. Monoclonal antibodies generated against BSA-coupled 402e accelerated the rate of cocaine hydrolysis by ~23,000x and eliminated the reinforcing effects of cocaine administration in rats.[30][31][32][33]
Structural/Functional intermediate analogues
Summarize
Perspective
Piperidine Analogues
- JZ-IV-10 (a "Modafinil hybrid" with nocaine.[36] cf. List of modafinil analogues)
A somewhat recent occurrence among tentative modern folklore which has traversed the circling of rumors mostly confined to the likes of universities and popular culture trivia has been that cocaine is one element, or molecule increment of weight or charge etc., away from the molecular structure of sugar.[37] Though such a statement is false as a general pretense, there is a dextrose based super-structure that has a vaguely similar overlay with cocaine which is "benzoyl-beta-D-glucoside."
Benztropine (3α-Diphenylmethoxy Tropane) Analogues
| Compound | S. Singh's alphanumeric assignation (name) |
R | R′ | Ki (nM) [3H]WIN 35428 binding |
IC50 (nM) [3H]DA uptake |
Selectivity
uptake/binding |
|---|---|---|---|---|---|---|
| (Cocaine) | 388 ± 47 | - | - | |||
| (GBR 12909) | 11.6 ± 31 | - | - | |||
 | ||||||
| (Benztropine) | H | H | 118 ± 9 | 403 ± 115 | 3.4 | |
| 249a | 4′-F | H | 32.2 ± 10 | 48 | 1.5 | |
| 249b (AHN 1-055) | 4′-F | 4′-F | 11.8 ± 1 | 71 | 6.0 | |
| 249c | 3′,4′-di-F | H | 27.9 ± 11 | 181 ± 45.7 | 6.5 | |
| 249d | 4′-Cl | H | 30.0 ± 12 | 115 | 3.8 | |
| 249e | 4′-Cl | 4′-Cl | 20.0 ± 14 | 75 | 3.8 | |
| 249f | 3′,4′-di-Cl | H | 21.1 ± 19 | 47 | 2.2 | |
| 249g | 3′,4′-di-Cl | F | 18.9 ± 14 | 24 | 1.3 | |
| 249h | 4′-Br | H | 37.9 ± 7 | 29 | 0.8 | |
| 249i | 4′-Br | 4′-Br | 91.6 | 34 | 0.4 | |
| 249j | 4′-NO2 | H | 197 ± 8 | 219 | 1.1 | |
| 249k | 4′-CN | H | 196 ± 9 | 222 | 1.1 | |
| 249l | 4′-CF3 | H | 635 ± 10 | 2155 | 3.4 | |
| 249m | 4′-OH | H | 297 ± 13 | 677 | 2.3 | |
| 249n | 4′-OMe | H | 78.4 ± 8 | 468 | 6.0 | |
| 249o | 4′-OMe | 4′-OMe | 2000 ± 7 | 2876 | 1.4 | |
| 249p | 4′-Me | H | 187 ± 5 | 512 | 2.7 | |
| 249q | 4′-Me | 4′-Me | 420 ± 7 | 2536 | 6.0 | |
| 249r | 4′-Et | H | 520 ± 8 | 984 | 1.9 | |
| 249s | 4′-t-Bu | H | 1918 | 4456 | 2.3 | |
| 250a | 3′-F | H | 68.5 ± 12 | 250 ± 64.7 | 3.6 | |
| 250b | 3′-F | 3′-F | 47.4 ± 1 | 407 ± 63.9 | 8.6 | |
| 250c | 3′-Cl | H | 21.6 ± 7 | 228 ± 77.1 | 10.5 | |
| 250d | 3′-CF3 | H | 187 ± 5 | 457 ± 72.0 | 2.4 | |
| 251a | 2′-F | H | 50.0 ± 12 | 140 ± 17.2 | 2.8 | |
| 251b | 2′-Cl | H | 228 ± 9 | 997 ± 109 | 4.4 | |
| 251c | 2′-Me | H | 309 ± 6 | 1200 ± 1.64 | 3.9 | |
| 251d | 2′-NH2 | H | 840 ± 8 | 373 ± 117 | 0.4 |
| Compound | S. Singh's alphanumeric assignation (name) |
R | R′ | IC50 (nM) DAT (Binding of [3H]WIN 35428) |
IC50 (nM) 5-HTT (Binding of [3H]Citalopram) |
Selectivity 5-HTT/DAT |
|---|---|---|---|---|---|---|
| (benztropine) | 312 ± 1.1 | 24100 ± 14800 | 77.2 | |||
| (WIN 35428) | 12.9 ± 1.1 | 160 ± 20 | 12.4 | |||
| R-256 | 2040 ± 283 | 1460 ± 255 | 0.7 | |||
 | ||||||
| S-257a | H | H | 33.5 ± 4.5 | 10100 ± 1740 | 301 | |
| S-257b | H | F | 13.2 ± 1.9 | 4930 ± 1200 | 373 | |
| S-257c (difluoropine) | F | F | 10.9 ± 1.2 | 3530 ± 1480 | 324 | |
| S-257d | H | Cl | 15.8 ± 0.95 | 5960 ± 467 | 377 | |
| S-257e | Cl | Cl | 91.4 ± 0.85 | 3360 ± 1480 | 36.8 | |
| S-257f | H | Br | 24.0 ± 4.6 | 5770 ± 493 | 240 | |
| S-257g | Br | Br | 72.0 ± 3.65 | 2430 ± 339 | 33.7 | |
| S-257h | H | I | 55.9 ± 10.3 | 9280 ± 1640 | 166 | |
| S-257i | Br | I | 389 ± 29.4 | 4930 ± 82 | 12.7 | |
| S-257j | I | I | 909 ± 79 | 8550 ± 442 | 9.4 | |
| S-257k | H | Me | 49.5 ± 6.0 | 13200 | 266 | |
| S-257l | Me | Me | 240 ± 18.4 | 9800 ± 2680 | 40.8 |
| Compound | S. Singh's alphanumeric assignation (name) |
R | n | IC50 (nM) DAT (Binding of [3H]WIN 35428) |
IC50 (nM) 5-HTT (Binding of [3H]Citalopram) |
Selectivity 5-HTT/DAT |
|---|---|---|---|---|---|---|
 | ||||||
| 258a | 20.3 ± 3.5 | - | - | |||
| 258b | H | 1 | 223 ± 53 | 4970 ± 700 | 22.3 | |
| 258c | H | 3 | 22.0 ± 11.9 | 19.7 ± 3 | 0.9 | |
| 258d | Br | 3 | 80.2 ± 8.8 | 234 ± 0.5 | 2.9 | |
| 258e | I | 3 | 119 ± 11 | 2200 ± 1250 | 18.5 | |
| 258f | H | 5 | 99.0 ± 28 | 550 ± 63 | 5.5 | |
| 259 | 616 ± 88 | 55200 ± 20000 | 89.3 |
| Compound | S. Singh's alphanumeric assignation (name) |
R | Ki (nM) DAT (Binding of [3H]WIN 35428) |
IC50 (nM) 5-HTT (Uptake of [3H]DA) |
Selectivity uptake/binding |
|---|---|---|---|---|---|
 | |||||
| 260 (AHN 2-003) | H | 11.2 ± 11 | 9.7 | 0.9 | |
| 261a | 3-phenylpropyl | 41.9 ± 11 | 230 | 5.5 | |
| 261b | indole-3-ethyl | 44.6 ± 11 | 1200 | 26.9 | |
| 261c | 4-phenylbutyl | 8.51 ± 14 | 39 | 4.6 | |
| 261d | 4-(4′-nitrophenyl)butyl | 20.2 ± 11 | 650 | 32.2 | |
| 261e | 3-(4′-fluorophenyl)propyl | 60.7 ± 12 | - | - | |
| 262a | n-butyl | 24.6 ± 8 | 370 | 15.0 | |
| 262b | cyclopropylmethyl | 32.4 ± 9 | 180 | 5.5 | |
| 262c | allyl | 29.9 ± 10 | 14 | 0.5 | |
| 262d | benzyl | 82.2 ± 15 | 290 | 3.5 | |
| 262e | 4-fluorobenzyl | 95.6 ± 10 | 200 | 2.1 | |
| 262f | cinnanyl | 86.4 ± 12 | 180 | 2.1 | |
| 262g | [bis(4-fluorophenyl)methoxy]ethyl | 634 ± 23 | - | - | |
| 262h | [(4-nitrophenyl)phenylmethoxy]ethyl | 57.0 ± 17 | - | - | |
| 263 | acetyl | 2340 | 4600 | 2.0 | |
| 264 | formyl | 2020 ± 13 | 5400 | 2.7 | |
| 265a | tosyl | 0%ɑ | - | - | |
| 265b | mesyl | 18%ɑ | - | - | |
| (AHN 2-005)[38] | CH2CH=CH2 | - | - | - | |
| (JHW 007)[38] | CH2CH2CH2CH3 | - | - | - | |
| (GA 2-99)[38] | CH2CH2NH2 | - | - | - | |
| (GA 103)[38] | CH2CH2CH2CH2Ph | - | - | - | |
 | 266 | 108 ± 12 | 130 | 1.2 | |
ɑInhibition at 10 μM
| Compound | S. Singh's alphanumeric assignation (name) |
IC50 (nM) DAT (Binding of [3H]WIN 35428) |
IC50 (nM) 5-HTT (Binding of [3H]Citalopram) | |
|---|---|---|---|---|
 | R/S-268 | 2β,3β | >10000 | >1660 |
| R/S-269 | 2α,3β | 20300 | >1660 | |
| R/S-270 | 2α,3α | 22300 | >1660 | |
 | R/S-271 | 2β,3α | 520 | >1660 |
| Benzatropine | Etybenzatropine | Difluoropine (O-620) | PG01053 | 276 | 277 |
|---|---|---|---|---|---|
 |  |  |  |  |  |
| MFZ 4-86 | MFZ 2-71 | 3-CPMT | JHW 007-d9[39] | GA 103 | AHN 1-055 |
 |  |  | 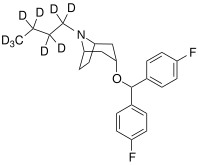 | 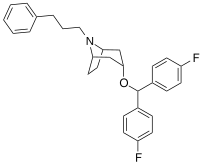 |  |
The binding of benztropine analogues to the DAT differs significantly from that of cocaine and the phenyltropanes. Benztropines are considered to be "atypical" DAT ligands because they stabilize the DAT in an inward-facing (closed-to-out) conformation, whereas cocaine and the phenyltropanes stabilize the DAT in an outward-facing (open-to-out) conformation. This difference in DAT binding may be responsible for the lack of cocaine-like behavioral effects observed in animal and human studies of the benztropine analogues and other “atypical” DAT inhibitors. [40] Studies of the structure-activity relationships of benztropine have shown that DAT affinity and selectivity over other monoamine transporters is enhanced by 4′,4′-difluorination. Modification of the tropane n-substituent was found to mitigate the anticholinergic effects of benztropine analogues by reducing M1 affinity.[41][42]
Tropanyl Isoxazoline Analogues
| 4a | 4c | 5a | 5c |
|---|---|---|---|
 |  |  |  |
| 6a | 6b | 6c | 7a |
 |  |  |  |
| 7b | 7c | 8a | 8b |
 |  |  |  |
| 8c | 9a | 9b | 9c |
 |  |  |  |
| 10a | 10b | 10c | 11a |
 |  |  |  |
| 11b | 12a | 12b | |
 |  |  |
Compound 7a (3′-methoxy-8-methyl-spiro(8-azabicyclo(3.2.1)octane-3,5′(4′H)-isoxazole) allosterically enhances SERT binding of other reuptake ligands. Compound 7a construed as a potentiating allosteric effect (by unveiling occluded configured serotonin uptake-area ligand-site on surface of transporter that allows for binding by exogenous ligand, when SERT is otherwise conformed in a transitional manner where a SERT ligand cannot bind, this effect with compound in question occurs) at concentrations of 10μM—30μM (wherein it acts by interconverting the conformational state of unexposed SERTs to ones exposing the SSRI binding site via a shift to the equilibrium of the MAT) while exerting an inhibitory orthosteric effect when concentrations reach >30μM and above.
7a is the only known compound to allosterically modulate SERT in such a way within in vitro conditions (tianeptine has been shown to do similar, but has only shown efficacy doing so in living in vivo tissue samples). Considering its noncompetitive inhibition of 5-HT transporters decreasing Vmax with small change in the Km for serotonin, putatively stabilizing the cytoplasm-facing conformation of SERT: in such respect it is considered to have the opposite effect profile of the anti-addiction drug ibogaine (save for the function by which its anti-addictive properties are thought to be mediated, i.e. α3β4 nicotinic channel blockage. cf. 18-Methoxycornaridine for such nicotinergic activity without the likewise SERT affinity).[44]
Compound 11a possesses similar effects, but acts on the DAT. Similarly, such peripheral DAT considerations (when, as often is, considered conformational rather than otherwise explained as being electrostatic) may constitute the difference in affinity, through allosertic occulsion, between cyclopentyl-ruthenium phenyltropane in its difference from the tricarbonyl-chromium
Alicyclic Amine Analogues
Dihydroimidazoles

See: List of Mazindol analogues
Mazindol is usually considered a non-habituating (in humans, and some other mammals, but is habituating for e.g. Beagles[av]) tetracyclic dopamine reuptake inhibitor (of somewhat spurious classification in the former).
It is a loosely functional analog used in cocaine research; due in large part to N-Ethylmaleimide being able to inhibit approximately 95% of the specific binding of [3H]Mazindol to the residues of the MAT binding site(s), however said effect of 10 mM N-Ethylmaleimide was prevented in its entirety by just 10 μM cocaine. Whereas neither 300 μM dopamine or D-amphetamine afforded sufficient protection to contrast the efficacy of cocaine.[aw]
Local anesthetics (not usually CNS stimulants)
Summarize
Perspective
Amylocaine, or Stovaine (above), the first synthetically constructed local anesthetic. Compare structure to dimethylaminopivalophenone (below), an analgesic (opioid). Cocaine's classification as a narcotic under U.S. legal code, as has been stretched to be medicinally rationalized such when defining terms very broadly (due to its topical numbing affect, hindering pain signals from CNS recognition via local anesthesia) usually considered an exaggeration of traditional medicine naming convention, in this instance between the first synthetic sodium channel blocker and one of the very simplest opioids there remains a measure of apparent structural similarity between the former anesthetic and latter analgesic "narcotic"; despite the highly differing methods of action for the respective 'pain-killing' properties of either.[45]
In animal studies, certain of the local anesthetics have displayed residual dopamine reuptake inhibitor properties,[46] although not normally ones that are easily available. These are expected to be more cardiotoxic than phenyltropanes. For example, dimethocaine has behavioral stimulant effects (and therefore not here listed below) if a dose of it is taken that is 10 times the amount of cocaine. Dimethocaine is equipotent to cocaine in terms of its anesthetic equivalency.[46] Intralipid "rescue" has been shown to reverse the cardiotoxic effects of sodium channel blockers and presumably those effects when from cocaine administered intravenously as well.
| Name | Other common names |
|---|---|
| Amylocaine | Stovaine |
| Articaine | Astracaine, Carticaine, Septanest, Septocaine, Ultracaine, Zorcaine |
| Benzocaine | Anbesol, Lanacane, Orajel |
| Bupivacaine | Marcaine, Sensorcaine, Vivacaine |
| Butacaine | Butyn |
| Chloroprocaine | Nesacaine |
| Cinchocaine/Dibucaine | Cincain, Cinchocaine, Nupercainal, Nupercaine, Sovcaine |
| Cyclomethycaine | Surfacaine, Topocaine |
| Etidocaine | Duranest |
| Eucaine | α-eucaine, β-eucaine |
| Fomocaine[47] | |
| Fotocaine[47] | |
| Hexylcaine | Cyclaine, Osmocaine |
| Levobupivacaine | Chirocaine |
| Lidocaine/Lignocaine | Xylocaine, Betacaine |
| Mepivacaine | Carbocaine, Polocaine |
| Meprylcaine/Oracaine | Epirocain |
| Metabutoxycaine | Primacaine |
| Phenacaine/Holocaine | Holocaine |
| Piperocaine | Metycaine |
| Pramocaine/Pramoxine | Pramoxine |
| Prilocaine | Citanest |
| Propoxycaine/Ravocaine | Pravocaine, Ranocaine, Blockain |
| Procaine/Novocaine | Borocaine (Procaine Borate), Ethocaine |
| Proparacaine/Alcaine | Alcaine |
| Quinisocaine | Dimethisoquin |
| Risocaine | Propaesin, Propazyl, Propylcain |
| Ropivacaine | Naropin |
| Tetracaine/Amethocaine | Pontocaine, Dicaine |
| Trimecaine | Mesdicain, Mesocain, Mesokain |
See also

- Coca alkaloids, the ones relating to cocaine biosynthesis include: benzoylecgonine, ecgonidine, ecgonine, hydroxytropacocaine, methylecgonine cinnamate, tropacocaine & truxilline
- Cocaine metabolites (Human), which include: benzoylecgonine (BE), ecgonine methyl ester (EME), ecgonine, norcocaine, p-hydroxycocaine, m-hydroxycocaine, p-hydroxybenzoylecgonine (pOHBE) & m-hydroxybenzoylecgonine
- Dopaminergics
- List of modafinil analogues and derivatives
- Federal Analog Act
- Pharmacophore
- Pharmacopoeia
- Pharmacokinetics
- Pharmacodynamics
Notes (inclu. specific locations of citations from within references used)
- [1] ←Page #969 (45th page of article) §III. ¶1. Final line. Last sentence.
- [1] ←Page #970 (46th page of article) Table 27. Figure 29.
- [1] ←Page #971 (47th page of article) Figure 30. & Page #973 (49th page of article) Table 28.
- [1] ←Page #971 (47th page of article) Figure 30 & Page #971 (47th page of article) Figure 30 & Page #973 (49th page of article) Table 28
- [1] ←Page #971 (47th page of article) Figure 30 & Page #971 (47th page of article) Figure 30 & Page #973 (49th page of article) Table 28
- [1] ←Page #972 (48th page of article) ¶2, Line 10.
- [1] ←Page #971 (47th page of article) Figure 30 & Page #971 (47th page of article) Figure 30 & Page #973 (49th page of article) Table 28
- [1] ←Page #971 (47th page of article) Figure 30 & Page #971 (47th page of article) Figure 30 & Page #973 (49th page of article) Table 28
- [1] ←Page #973 (49th page of article) §C. & Page #974 (50th page of article) Figure 31 & Page #976 (52nd page of article) Table 29.
- [1] ←Page #974 (50th page of article) Final ¶ (5th), Second line.
- [1] ←Page #975 (51st page of article) First ¶, first line.
- [1] ←Page #975 (51st page of article) First ¶, 4th line.
- [1] ←Page #974 (50st page of article) First (left) column, third ¶
- [1] ←Page #937 (13th page of article) Second (right) column, first ¶. Above/before §2
- [1] ←Page #974 (50st page of article) First (left) column, fourth ¶
- [1] ←Page #974 (50th page of article) Figure 31 & Page #977 (53rd page of article) Table 30.
- [1] ←Page #974 (50th page of article) Figure 31 & Page #977 (53rd page of article) Table 30.
- [1] ←Page #974 (50th page of article) Figure 31 & Page #977 (53rd page of article) Table 30.
- [1] ←Page #974 (50th page of article) Figure 31 & Page #977 (53rd page of article) Table 30.
- [1] ←Page #978 (54th page of article) §D & Page #980 (56th page of article) Figure 33 & Page #981 (57th page of article) Table 32.
- [1] ←Page #964 (39th page of article) Table 23.
- [1] ←Page #980 (56th page of article) Scheme 52.
- [1] ←Page #963 (39th page of article) 2nd (right side) column, 2nd paragraph.
- [1] ←Page #967 (43rd page of article) §C. & Page #967 (43rd page of article) Table 25
- [1] ←Page #982 (58th page of article) §G & Page #983 (59th page of article) Figure 36 & Page #984 (60th page of article) Table 35.
- [1] ←Page #979 (55th page of article) Table 31.
- [1] ←Page #981 (57th page of article) §E & Page #982 (58th page of article) Table 33.
- [1] ←Page #970 (46th page of article) §B, 10th line
- [1] ←Page #971 (47th page of article) 1st ¶, 10th line
- [1] ←Page #949 (25th page of article) 3rd ¶, 20th line
- [1] ←Page #982 (58th page of article) §F, Table 34 & Figure 35.
- [1] ←Page #982 (58th page of article) 3rd ¶, lines 2, 5 & 6.
- [1] ←Page #984 (60th page of article) Figure 37.
- [1] ←Page #984 (60th page of article) §H, Figure 37 & Page #985 (61st page of article) Table 36.
- [1] ←Page #984 (60th page of article) Scheme 56.
- [1] ←Page #986 (62nd page of article) §I, Table 37 & Scheme 58
- [1] ←Page #1,014 (90th page of article) §VIII, A. Figure 59.
- [1] ←Page #1,016 (92nd page of article) Figure 60.
- [1] ←Page #987 (63rd page of article) §IV, Figure 39 & Page #988 (64th page of article) Table 38.
- [1] ←Page #987 (63rd page of article) Figure 40, Page #988 (64th page of article) §B & Page #989 (65th page of article) Table 39.
- [1] ←Page #987 (63rd page of article) Figure 41, Page #989 (65th page of article) §C & Page #990 (66th page of article) Table 40.
- [1] ←Page #988 (64th page of article) Figure 42, Page #990 (66th page of article) §2 & Page #992 (68th page of article) Table 41.
- [1] ←Page #988 (64th page of article) Figure 43, Page #992 (68th page of article) §3 & Table 42.
- [1] ←Page #1,011 (87th page of article) §VII (7) 1st ¶.
- [1] ←Page #969 (45th page of article) 2nd (right-side) column 2nd ¶.
References
External links
Wikiwand - on
Seamless Wikipedia browsing. On steroids.