Histone H2A type 1 is a protein that in humans is encoded by the HIST1H2AL gene.[5][6][7][8]
Quick Facts H2AC16, Available structures ...
| H2AC16 |
|---|
 |
|
| Identifiers |
|---|
| Aliases | H2AC16, H2A.i, H2A/i, H2AFI, dJ193B12.9, histone cluster 1, H2al, histone cluster 1 H2A family member l, H2A clustered histone 16, HIST1H2AL, H2AC11, H2AC15, H2AC17, H2AC13 |
|---|
| External IDs | OMIM: 602793; MGI: 2448290; HomoloGene: 136768; GeneCards: H2AC16; OMA:H2AC16 - orthologs |
|---|
|
|
|
|
|
| Wikidata |
|
Close
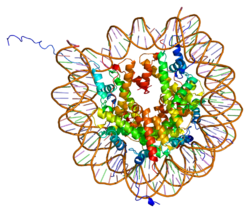
Histones are basic nuclear proteins that are responsible for the nucleosome structure of the chromosomal fiber in eukaryotes. Two molecules of each of the four core histones (H2A, H2B, H3, and H4) form an octamer, around which approximately 146 bp of DNA is wrapped in repeating units, called nucleosomes. The linker histone, H1, interacts with linker DNA between nucleosomes and functions in the compaction of chromatin into higher order structures. This gene is intronless and encodes a member of the histone H2A family. Transcripts from this gene lack polyA tails but instead contain a palindromic termination element. This gene is found in the small histone gene cluster on chromosome 6p22-p21.3.[8]
- Du Clos TW, Zlock LT, Marnell L (1991). "Definition of a C-reactive protein binding determinant on histones". J. Biol. Chem. 266 (4): 2167–71. doi:10.1016/S0021-9258(18)52224-9. PMID 1989977.
- El Kharroubi A, Piras G, Zensen R, Martin MA (1998). "Transcriptional activation of the integrated chromatin-associated human immunodeficiency virus type 1 promoter". Mol. Cell. Biol. 18 (5): 2535–44. doi:10.1128/mcb.18.5.2535. PMC 110633. PMID 9566873.
- Zhang Y, Sun ZW, Iratni R, et al. (1998). "SAP30, a novel protein conserved between human and yeast, is a component of a histone deacetylase complex". Mol. Cell. 1 (7): 1021–31. doi:10.1016/S1097-2765(00)80102-1. PMID 9651585.
- Carrier F, Georgel PT, Pourquier P, et al. (1999). "Gadd45, a p53-responsive stress protein, modifies DNA accessibility on damaged chromatin". Mol. Cell. Biol. 19 (3): 1673–85. doi:10.1128/MCB.19.3.1673. PMC 83961. PMID 10022855.
- Albig W, Trappe R, Kardalinou E, et al. (1999). "The human H2A and H2B histone gene complement". Biol. Chem. 380 (1): 7–18. doi:10.1515/BC.1999.002. PMID 10064132. S2CID 12807248.
- Deng L, de la Fuente C, Fu P, et al. (2001). "Acetylation of HIV-1 Tat by CBP/P300 increases transcription of integrated HIV-1 genome and enhances binding to core histones". Virology. 277 (2): 278–95. doi:10.1006/viro.2000.0593. PMID 11080476.
- Deng L, Wang D, de la Fuente C, et al. (2001). "Enhancement of the p300 HAT activity by HIV-1 Tat on chromatin DNA". Virology. 289 (2): 312–26. doi:10.1006/viro.2001.1129. PMID 11689053.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. Bibcode:2002PNAS...9916899M. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932.
- Kzhyshkowska J, Rusch A, Wolf H, Dobner T (2003). "Regulation of transcription by the heterogeneous nuclear ribonucleoprotein E1B-AP5 is mediated by complex formation with the novel bromodomain-containing protein BRD7". Biochem. J. 371 (Pt 2): 385–93. doi:10.1042/BJ20021281. PMC 1223277. PMID 12489984.
- Lusic M, Marcello A, Cereseto A, Giacca M (2004). "Regulation of HIV-1 gene expression by histone acetylation and factor recruitment at the LTR promoter". EMBO J. 22 (24): 6550–61. doi:10.1093/emboj/cdg631. PMC 291826. PMID 14657027.