Top Qs
Timeline
Chat
Perspective
Copper(II) bromide
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
Copper(II) bromide (CuBr2) is a chemical compound that forms an unstable tetrahydrate CuBr2·4H2O. It is used in photographic processing as an intensifier and as a brominating agent in organic synthesis.[2]
It is also used in the copper vapor laser, a class of laser where the medium is copper bromide vapour formed in-situ from hydrogen bromide reacting with the copper discharge tube.[3] Producing yellow or green light, it is used in dermatological applications.
Remove ads
Synthesis
Copper(II) bromide can be obtained by combining copper oxide and hydrobromic acid:[4]
- CuO + 2HBr → CuBr2 + H2O.
The tetrahydrate can be produced by recrystallization of solutions of copper(II) bromide at 0 °C. If heated above 18 °C, it releases water to produce the anhydrous form.[5]
Purification
Copper(II) bromide is purified by crystallization twice from water, filtration to remove any CuBr and concentration under vacuum. This product is dehydrated using phosphorus pentoxide.[6]
Molecular and crystal structure
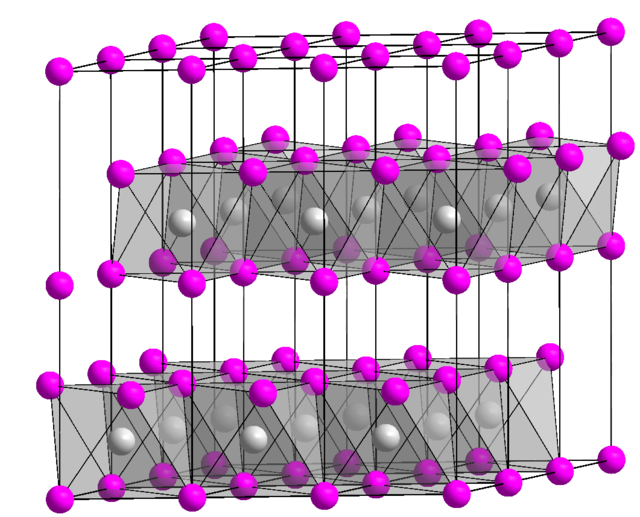
In the solid state CuBr2 has a polymeric structure, with CuBr4 planar units connected on opposite sides to form chains. The crystal structure is monoclinic, space group C2/m, with lattice constants a = 714 pm, b = 346 pm, c = 718 pm, e ß = 121° 15'.[7] CuBr2 monomeric units are present in the gas phase at high temperature.[8]
The tetrahydrate, structurally formulated as [CuBr2(H2O)2]·2H2O, has a monoclinic crystal structure and consists of distorted square planartrans-[CuBr2(H2O)2] centres as well as two molecules of water.[5]
Reactions
Copper(II) bromide in chloroform-ethyl acetate reacts with ketones resulting in the formation of alpha-bromo ketones. The resulting product can be directly used for the preparation of derivatives. This heterogeneous method is reported to be the most selective and direct method of formation of α-bromo ketones.[9]
Dibromination of NPGs, n-pentenyl glycosides, using CuBr2/LiBr reagent combination was performed in order for an NPG to serve as a glycosyl acceptor during halonium-promoted couplings. Such reaction gives high yield of the dibromides from alkenyl sugars that are resistant to a direct reaction with molecular bromine.[10]
Remove ads
Usage
Copper(II) bromide lasers produce pulsed yellow and green light and have been studied as a possible treatment for cutaneous lesions.[11] Experiments have also shown copper bromide treatment to be beneficial for skin rejuvenation.[12] It has been widely used in photography as its solution was used as the bleaching step for intensifying collodion and gelatin negatives.[13] Copper(II) bromide has also been proposed as a possible material in humidity indicator cards.[14]
Remove ads
Safety
Copper(II) bromide is harmful if swallowed. It affects the central nervous system, brain, eyes, liver, and kidneys. It causes irritation to skin, eyes, and respiratory tract.
Natural occurrence
As of 2020[update], pure copper(II) bromide is unknown among minerals. However, barlowite, Cu4BrF(OH)6, contains both copper and bromide.[15][16]
See also
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads



