Clostridium sporogenes
Species of bacterium From Wikipedia, the free encyclopedia
Clostridium sporogenes is a species of Gram-positive bacteria that belongs to the genus Clostridium. Like other strains of Clostridium, it is an anaerobic, rod-shaped bacterium that produces oval, subterminal endospores[2] and is commonly found in soil. Unlike Clostridium botulinum, it does not produce the botulinum neurotoxins. In colonized animals, it has a mutualistic rather than pathogenic interaction with the host.
| Clostridium sporogenes | |
|---|---|
 | |
| Scientific classification | |
| Domain: | Bacteria |
| Kingdom: | Bacillati |
| Phylum: | Bacillota |
| Class: | Clostridia |
| Order: | Eubacteriales |
| Family: | Clostridiaceae |
| Genus: | Clostridium |
| Species: | C. sporogenes |
| Binomial name | |
| Clostridium sporogenes (Metchnikoff 1908) Bergey et al. 1923[1] | |
It is being investigated as a way to deliver cancer-treating drugs to tumours in patients.[3] C. sporogenes is often used as a surrogate for C. botulinum when testing the efficacy of commercial sterilisation.[4]
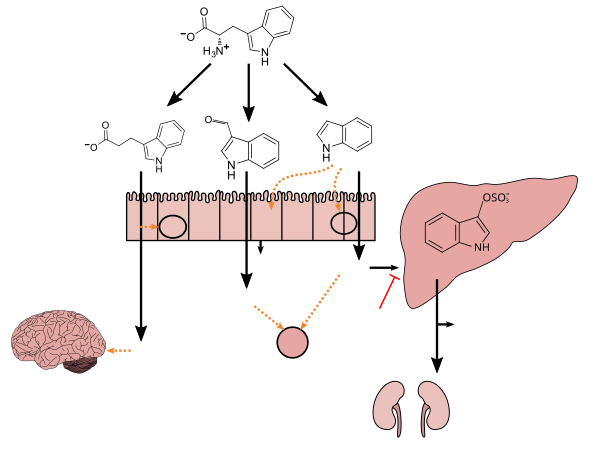
Clostridium sporogenes colonizes the human gastrointestinal tract, but is only present in a subset of the population; in the intestine, it uses tryptophan to synthesize indole and subsequently 3-indolepropionic acid (IPA)[5] – a type of auxin (plant hormone)[6][7] – which serves as a potent neuroprotective antioxidant within the human body and brain.[5][8][9][10] IPA is an even more potent scavenger of hydroxyl radicals than melatonin.[8][9][10] Similar to melatonin but unlike other antioxidants, it scavenges radicals without subsequently generating reactive and pro-oxidant intermediate compounds.[8][9][11] C. sporogenes is the only species of bacteria known to synthesize 3-indolepropionic acid in vivo at levels which are subsequently detectable in the blood stream of the host.[5][12]
Tryptophan metabolism by human gut microbiota ()
|
References
External links
Wikiwand - on
Seamless Wikipedia browsing. On steroids.