Top Qs
Timeline
Chat
Perspective
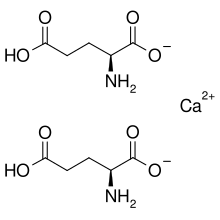
Calcium diglutamate
Chemical compound From Wikipedia, the free encyclopedia
Remove ads
Calcium diglutamate, sometimes abbreviated CDG and also called calcium biglutamate, is a compound with formula Ca(C5H8NO4)2. It is a calcium acid salt of glutamic acid. CDG is a flavor enhancer (E number E623)—it is the calcium analog of monosodium glutamate (MSG). Because the glutamate is the actual flavor-enhancer, CDG has the same flavor-enhancing properties as MSG but without the increased sodium content.[1] Notably, only the L isomer is used in flavouring as D-glutamate does not have an umami/savoury flavour.[2][3]
As a soluble source of calcium ions, this chemical is also used as a first-aid treatment for exposure to hydrofluoric acid.[4]
Remove ads
Synthesis and reactions
Calcium di-glutamate can be prepared by reacting calcium carbonate with two molar equivalents of glutamic acid:[5]
- CaCO3 + 2 HOOC(CH2)2CH(NH2)COOH → Ca(OOC(CH2)2CH(NH3)COO)2 + H2O + CO2↑
Concentration of the solution to a syrup under reduced pressure, followed by gradual crystallisation, affords the monohydrate.[5] Structurally, the glutamate anion is zwitterionic, with the amino group protonated (pKa = 9.47) and both carboxylic-acid groups (pKa = 2.10, 4.07) in their deprotonated carboxylate form.[6]
Calcium di-L-glutamate can be used to prepare other glutamates through metathesis with a soluble sulfate, carbonate or hydroxide salt. For example, manganese(II) di-L-glutamate can be prepared through metathesis with manganese(II) sulphate:[7]
- Ca(OOC(CH2)2CH(NH3)COO)2 + MnSO4 → Mn(OOC(CH2)2CH(NH3)COO)2 + CaSO4↓
Remove ads
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.
Remove ads