Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
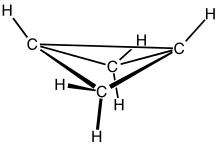
Bicyclo[1.1.0]butane is an organic compound with the formula C4H6. It is a bicyclic molecule consisting of two cis-fused cyclopropane rings, and is a colorless and easily condensed gas.[1] Bicyclobutane is noted for being one of the most strained compounds that is isolatable on a large scale — its strain energy is estimated at 63.9 kcal mol−1. It is a nonplanar molecule, with a dihedral angle between the two cyclopropane rings of 123°.[2]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Bicyclo[1.1.0]butane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H6 | |
| Molar mass | 54.092 g·mol−1 |
| Appearance | colorless gas |
| Boiling point | 8.3 ± 0.2 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The first reported bicyclobutane was the ethyl carboxylate derivative, C4H5CO2Et, which was prepared by dehydrohalogenation the corresponding bromocyclobutanecarboxylate ester with sodium hydride.[2] The parent hydrocarbon was prepared from 1-bromo-3-chlorocyclobutane by conversion of the bromocyclobutanecarboxylate ester,[1] followed by intramolecular Wurtz coupling using molten sodium.[3] The intermediate 1-bromo-3-chlorocyclobutane can also be prepared via a modified Hunsdiecker reaction from 3-chlorocyclobutanecarboxylic acid using mercuric oxide and bromine:[4]
A synthetic approach to bicyclobutane derivatives involves ring closure of a suitably substituted 2-bromo-1-(chloromethyl)cyclopropane with magnesium in THF.[5] Substituted bicyclo[1.1.0]butanes can also be prepared from the reaction of iodo-bicyclo[1.1.1]pentanes with amines, thiols, and sulfinate salts.[6] Bicyclo[1.1.0]butanes are explored in medicinal chemistry as covalent reactive groups.[7]
Stereochemical evidence indicates that bicyclobutane undergoes thermolysis to form 1,3-butadiene with an activation energy of 41 kcal mol−1 via a concerted pericyclic mechanism (cycloelimination, [σ2s+σ2a]).[8]
Linolenic acid can be converted into its bicyclobutane derivative using a fusion protein produced by a strain of the cyanobacterium Anabaena sphaerica (strain PCC 7120).[9] The other group reported a directed evolution approach, whereby engineered heme protein was expressed in E. coli and optimized for rate and yield of a substituted bicyclobutane derivative.[10]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.