
Zingibain
Cysteine protease enzyme / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Zingibain?
Summarize this article for a 10 year old
Zingibain, zingipain, or ginger protease (EC 3.4.22.67) is a cysteine protease enzyme found in ginger (Zingiber officinale) rhizomes.[1][2][3] It catalyses the preferential cleavage of peptides with a proline residue at the P2 position. It has two distinct forms, ginger protease I (GP-I) and ginger protease II (GP-II).[4]
| Zingibain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
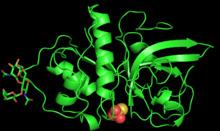 PYMOL generated 3D structure of zingibain monomer | |||||||||
| Identifiers | |||||||||
| EC no. | 3.4.22.67 | ||||||||
| CAS no. | 246044-91-7 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
As a member of the papain-like protease family of cysteine proteases, zingibain shares several structural and functional similarities with more well-studied enzymes such as papain, bromelain, and actinidin. These peptidases contain an active cysteine residue in their centers that catalyzes the hydrolytic cleavage of peptide bonds. Zingibain is noted for its activity as a proteinase and a collagenase.[1]
It was first isolated, purified, and reported in 1973 by Ichikawa et al. at Japan Women's University.[5] Recently, zingibain was found to exist as two isozymes, GP-I and GP-II, which were isolated by chromatography, with molecular weights of approximately 22,500 Da.[5]