Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
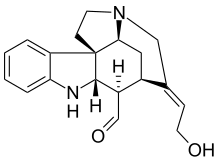
The so-called Wieland-Gumlich aldehyde (6) is an indoline derived by chemical degradation from strychnine. This compound is of some commercial interest as a chemical intermediate. It was first synthesized in 4 steps from strychnine (1)[2][3][4] by Walter Gumlich and Koozoo Kaziro working in the laboratory of Heinrich Wieland. This degradation study was part of an attempt to elucidate the chemical structure of strychnine.
 | |
| Names | |
|---|---|
| IUPAC name
(1S,9S,10R,11R,12E,17S)-12-(2-hydroxyethylidene)-8,14-diazapentacyclo[9.5.2.01,9.02,7.014,17]octadeca-2,4,6-triene-10-carbaldehyde | |
| Other names
Caracurine VII, Deacetyldiaboline | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| UNII |
|
| |
| |
| Properties | |
| C19H22N2O2 | |
| Molar mass | 310.397 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
This degradation takes place through conversion of strychnine to the oxime 2 using amyl nitrite, Beckmann fragmentation of 2 to the carbamic acid 3 by use of thionyl chloride, decarboxylation of 3 to nitrile 4, and nucleophilic displacement of cyanide by barium hydroxide to give hemiacetal 5, which is in equilibrium with the Wieland-Gumlich aldehyde (6).
The Wieland-Gumlich aldehyde reverts to strychnine in a single reaction using malonic acid, acetic anhydride and sodium acetate in acetic acid.[5]
The Wieland-Gumlich aldehyde has been used in the industrial synthesis of alcuronium chloride (Alloferin) via dimerization.[6]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.