Loading AI tools
Vanadium(IV) fluoride (VF4) is an inorganic compound of vanadium and fluorine. It is paramagnetic yellow-brown solid that is very hygroscopic.[2] Unlike the corresponding vanadium tetrachloride, the tetrafluoride is not volatile because it adopts a polymeric structure.[5] It decomposes before melting.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
vanadium tetrafluoride | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
| ECHA InfoCard | 100.030.143 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
| UN number | UN2923 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| F4V | |||
| Molar mass | 126.9351 g·mol−1 | ||
| Appearance | Lime green powder, hygroscopic[1] | ||
| Odor | Odorless | ||
| Density | 3.15 g/cm3 (20 °C)[1] 2.975 g/cm3 (23 °C)[2] | ||
| Melting point | 325 °C (617 °F; 598 K) at 760 mmHg decomposes[1] | ||
| Boiling point | Sublimes[1] | ||
| Very soluble[1] | |||
| Solubility | Soluble in acetone, acetic acid Very slightly soluble in SO2Cl2, alcohols, CHCl3[2] | ||
| Structure | |||
| Monoclinic, mP10 | |||
| P21/c, No. 14 | |||
| Thermochemistry | |||
Std molar entropy (S⦵298) |
126 J/mol·K[3] | ||
Std enthalpy of formation (ΔfH⦵298) |
−1412 kJ/mol[3] | ||
Gibbs free energy (ΔfG⦵) |
−1312 kJ/mol[3] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Eye hazards |
Causes serious damage | ||
Skin hazards |
Causes burns | ||
| GHS labelling:[4] | |||
  | |||
| Danger | |||
| H300, H314, H330 | |||
| P260, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P320, P330, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
VF4 can be prepared by treating VCl4 with HF:
- VCl4 + 4 HF → VF4 + 4 HCl
It was first prepared in this way.[6]
It decomposes at 325 °C, undergoing disproportionation to the tri- and pentafluorides:[2]
- 2 VF4 → VF3 + VF5
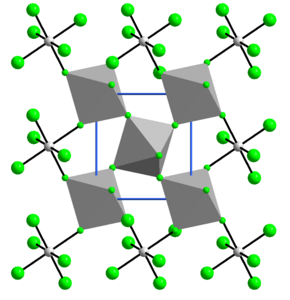
The structure of VF4 is related to that of SnF4. Each vanadium centre is octahedral, surrounded by six fluoride ligands. Four of the fluoride centers bridge to adjacent vanadium centres.[7]
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.