User:Mr. Ibrahem/Dacarbazine
From Wikipedia, the free encyclopedia
Dacarbazine (DTIC), also known as imidazole carboxamide, is a chemotherapy medication used in the treatment of melanoma and Hodgkin's lymphoma.[1] For Hodgkin's it is often used together with vinblastine, bleomycin, and doxorubicin.[1] It is given by injection into a vein.[1]
 | |
| Clinical data | |
|---|---|
| Pronunciation | /dəˈkɑːrbəˌziːn/ |
| Trade names | DTIC-Dome, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682750 |
| Routes of administration | IV |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 100% (IV) |
| Metabolism | Extensive |
| Elimination half-life | 5 hours |
| Excretion | Kidney (40% as unchanged dacarbazine) |
| Identifiers | |
| |
| Chemical and physical data | |
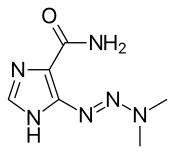
| Formula | C6H10N6O |
| Molar mass | 182.187 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include loss of appetite, vomiting, low white blood cell count, and low platelets.[1] Other serious side effects include liver problems and allergic reactions.[1] It is unclear if use in pregnancy is safe for the baby.[1] Dacarbazine is in the alkylating agent and purine analog families of medication.[1]
Dacarbazine was approved for medical use in the United States in 1975.[1] It is on the World Health Organization's List of Essential Medicines.[2] The wholesale cost in the developing world is about US$7.45–18.24 per 200 mg vial.[3] In the United Kingdom this dose costs the NHS about 7.50 pounds.[4]