Triethylamine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Triethylamine?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with Triethanolamine.
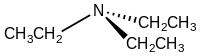
Triethylamine is the chemical compound with the formula N(CH2CH3)3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine or tetraethylammonium, for which TEA is also a common abbreviation.[8][9] It is a colourless volatile liquid with a strong fishy odor reminiscent of ammonia. Like diisopropylethylamine (Hünig's base), triethylamine is commonly employed in organic synthesis, usually as a base.
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
N,N-Diethylethanamine | |||
| Other names
(Triethyl)amine Triethylamine (no longer IUPAC name[1]) | |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| Abbreviations | TEA[2] | ||
| 605283 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.004.064 | ||
| EC Number |
| ||
| KEGG |
| ||
| MeSH | triethylamine | ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1296 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties[3] | |||
| C6H15N | |||
| Molar mass | 101.193 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Odor | Fishy, ammoniacal | ||
| Density | 0.7255 g mL−1 | ||
| Melting point | −114.70 °C; −174.46 °F; 158.45 K | ||
| Boiling point | 88.6 to 89.8 °C; 191.4 to 193.5 °F; 361.7 to 362.9 K | ||
| 112.4 g/L at 20 °C[4] | |||
| Solubility | miscible with organic solvents | ||
| log P | 1.647 | ||
| Vapor pressure | 6.899–8.506 kPa | ||
Henry's law constant (kH) |
66 μmol Pa−1 kg−1 | ||
| Acidity (pKa) | 10.75 (for the conjugate acid) (H2O), 9.00 (DMSO)[5] | ||
| -81.4·10−6 cm3/mol | |||
Refractive index (nD) |
1.401 | ||
| Thermochemistry | |||
Heat capacity (C) |
216.43 J K−1 mol−1 | ||
Std enthalpy of formation (ΔfH⦵298) |
−169 kJ mol−1 | ||
Std enthalpy of combustion (ΔcH⦵298) |
−4.37763 to −4.37655 MJ mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
   | |||
| Danger | |||
| H225, H302, H312, H314, H332 | |||
| P210, P280, P305+P351+P338, P310 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −15 °C (5 °F; 258 K) | ||
| 312 °C (594 °F; 585 K) | |||
| Explosive limits | 1.2–8% | ||
Threshold limit value (TLV) |
2 ppm (8 mg/m3) (TWA), 4 ppm (17 mg/m3) (STEL) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
| ||
LCLo (lowest published) |
1425 ppm (mouse, 2 hr)[6] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 25 ppm (100 mg/m3)[7] | ||
REL (Recommended) |
None established[7] | ||
IDLH (Immediate danger) |
200 ppm[7] | ||
| Related compounds | |||
Related amines |
|||
Related compounds |
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close


