Thujone
Group of four possible stereoisomers found in various plants: a.o., absinthe and mint / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Thujone?
Summarize this article for a 10 year old
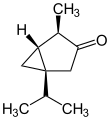
Thujone (/ˈθuːdʒoʊn/ ⓘ[2]) is a ketone and a monoterpene that occurs predominantly in two diastereomeric (epimeric) forms: (−)-α-thujone and (+)-β-thujone.[3][4]
| |||
 Ball-and-stick model of (−)-α-thujone[1] | |||
| Names | |||
|---|---|---|---|
| IUPAC names
α: (1S,4R,5R)-4-Methyl-1-(propan-2-yl)bicyclo[3.1.0]hexan-3-one β: (1S,4S,5R)-4-methyl-1-propan-2-ylbicyclo[3.1.0]hexan-3-one | |||
| Other names
Bicyclo[3.1.0]hexan-3-one, 4-methyl-1-(1-methylethyl)-, [1S-(1α,4α,5α)]- α-Thujone β-Thujone Thujone, cis 3-Thujanone, (1S,4R,5R)-(−)- Thujon 3-Thujanone, (−)- l-Thujone; 4-Methyl-1-(1-methylethyl)bicyclo[3.1.0]hexan-3-one-, (1S,4R,5R)- 3-Thujone; cis-Thujone (Z)-Thujone (−)-Thujone; Bicyclo(3.1.0)hexan-3-one, 4-methyl-1-(1-methylethyl)-, (1S,4R,5R)- NSC 93742 1-isopropyl-4-methylbicyclo[3.1.0]hexan-3-one | |||
| Identifiers | |||
| |||
3D model (JSmol) |
| ||
| 4660369 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.013.096 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID |
|||
| UNII |
| ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C10H16O | |||
| Molar mass | 152.237 g·mol−1 | ||
| Density | 0.92 g/cm3 (β-thujone); 0.9116 g/cm3 (α-thujone) | ||
| Melting point | <25 °C | ||
| Boiling point | 203 °C (397 °F; 476 K) (alpha,beta-thujone) | ||
| 407 mg/L | |||
| Hazards | |||
| GHS labelling: | |||
 | |||
| Warning | |||
| H302 | |||
| P264, P270, P301+P312, P330, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Though it is best known as a chemical compound in the spirit absinthe, it is unlikely to be responsible for absinthe's alleged stimulant and psychoactive effects due to the small quantities present.[5][6][7]
Thujone acts on the GABAA receptor as an antagonist. As a competitive antagonist of GABAA receptor, thujone alone is considered to be convulsant,[8] though by interfering with the inhibitory transmitter GABA, it may convey stimulating, mood-elevating effects at low doses.[citation needed] It is also found in perfumery as a component of several essential oils.[citation needed]
In addition to the naturally occurring (−)-α-thujone and (+)-β-thujone, two other forms are possible: (+)-α-thujone and (−)-β-thujone. In 2016, they were found in nature as well,[9] in Salvia officinalis.
- (−)-α-thujone
- (+)-α-thujone
- (+)-β-thujone
- (−)-β-thujone