Thiosulfurous acid
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Thiosulfite?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
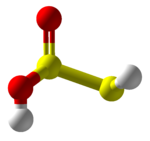
Thiosulfurous acid is a hypothetical chemical compound with the formula HS−S(=O)−OH or HO−S(=S)−OH. Attempted synthesis leads to polymers.[3] It is a low oxidation state (+1) sulfur acid.[4] It is the Arrhenius acid for disulfur monoxide. Salts derived from thiosulfurous acid, which are also unknown, are named "thiosulfites", "thionosulfites" or "sulfurothioites". The ion is S=SO2−
2.
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names | |||
| Systematic IUPAC name
hydroxidooxidosulfanidosulfur[1] | |||
| Other names
sulfurothionous acid Thiothionyl hydroxide (minor tautomer) | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| 184467 | |||
PubChem CID |
|||
| |||
| |||
| Properties | |||
| H2S2O2 | |||
| Molar mass | 98.14668 | ||
| Conjugate base | Thiosulfite | ||
| Related compounds | |||
Related compounds |
thiosulfuric acid SSO thiosulfinic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close