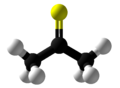
Thioacetone
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Thioacetone?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Thioacetone is an organosulfur compound belonging to the -thione group called thioketones with a chemical formula (CH3)2CS. It is an unstable orange or brown substance that can be isolated only at low temperatures.[3] Above −20 °C (−4 °F), thioacetone readily converts to a polymer and a trimer, trithioacetone.[4] It has an extremely potent, unpleasant odor, and is considered one of the worst-smelling chemicals known to humanity.[5]
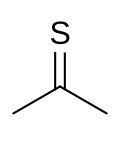
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Propane-2-thione[1] | |||
| Systematic IUPAC name
Thiopropan-2-one | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C3H6S | |||
| Molar mass | 74.14 g·mol−1 | ||
| Appearance | Orange to brown liquid | ||
| Odor | Intensely sulfurous, leak-like | ||
| Melting point | −55°C(218.15k/-67°F)[2] | ||
| Boiling point | 70°C(343.15k/158°F)[2] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Odor, skin irritant | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
Thioacetone was first obtained in 1889 by Baumann and Fromm, as a minor impurity in their synthesis of trithioacetone.[2]