Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
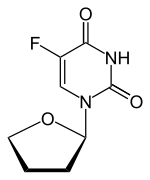
Tegafur is a chemotherapeutic prodrug of 5-fluorouracil (5-FU) used in the treatment of cancers. It is a component of the combination drug tegafur/uracil. When metabolised, it becomes 5-FU.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Other names | 5-fluoro-1-(oxolan-2-yl)pyrimidine-2,4-dione |
| AHFS/Drugs.com | International Drug Names |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 3.9-11 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.038.027 |
| Chemical and physical data | |
| Formula | C8H9FN2O3 |
| Molar mass | 200.169 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
It was patented in 1967 and approved for medical use in 1972.[2]
As a prodrug to 5-FU it is used in the treatment of the following cancers:[3]
It is often given in combination with drugs that alter its bioavailability and toxicity such as gimeracil, oteracil or uracil.[3] These agents achieve this by inhibiting the enzyme dihydropyrimidine dehydrogenase (uracil/gimeracil) or orotate phosphoribosyltransferase (oteracil).[3]
The major side effects of tegafur are similar to fluorouracil and include myelosuppression, central neurotoxicity and gastrointestinal toxicity (especially diarrhoea).[3] Gastrointestinal toxicity is the dose-limiting side effect of tegafur.[3] Central neurotoxicity is more common with tegafur than with fluorouracil.[3]
The dihydropyrimidine dehydrogenase (DPD) enzyme is responsible for the detoxifying metabolism of fluoropyrimidines, a class of drugs that includes 5-fluorouracil, capecitabine, and tegafur.[5] Genetic variations within the DPD gene (DPYD) can lead to reduced or absent DPD activity, and individuals who are heterozygous or homozygous for these variations may have partial or complete DPD deficiency; an estimated 0.2% of individuals have complete DPD deficiency.[5][6] Those with partial or complete DPD deficiency have a significantly increased risk of severe or even fatal drug toxicities when treated with fluoropyrimidines; examples of toxicities include myelosuppression, neurotoxicity and hand-foot syndrome.[5][6]
It is a prodrug to 5-FU, which is a thymidylate synthase inhibitor.[3]
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.