
Sulforaphane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sulforaphane?
Summarize this article for a 10 year old
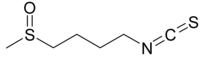
Sulforaphane (sometimes sulphoraphane in British English) is a compound within the isothiocyanate group of organosulfur compounds.[1] It is produced when the enzyme myrosinase transforms glucoraphanin, a glucosinolate, into sulforaphane upon damage to the plant (such as from chewing or chopping during food preparation), which allows the two compounds to mix and react.
 | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Isothiocyanato-4-(methanesulfinyl)butane | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H11NOS2 | |
| Molar mass | 177.29 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sulforaphane is present in cruciferous vegetables, such as broccoli, Brussels sprouts, and cabbage.[1]
Sulforaphane has two possible stereoisomers due to the presence of a stereogenic sulfur atom.[2]
The R-sulforaphane enantiomer occurs naturally, while the S-sulforaphane can be synthesized.[3]
 Glucoraphanin, the glucosinolate precursor to sulforaphane |