Sulfilimine
Class of chemical compounds containing an S=N bond / From Wikipedia, the free encyclopedia
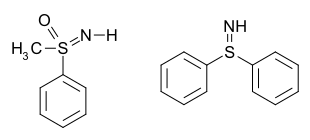
In chemistry, a sulfilimine (or sulfimide) is a type of chemical compound containing a sulfur-to-nitrogen bond which is often represented as a double bond (S=N). In fact, a double bond violates the octet rule, and the bond may be considered a single bond with a formal charge of +1 on the sulfur and a formal charge of −1 on the nitrogen. The parent compound is sulfilimine H2S=NH, which is mainly of theoretical interest.

Examples include S,S-diphenylsulfilimine[2] and sulfoximines [Category] such as methylphenylsulfoximine:[3] In the case of a sulfoximine, the bonds can be considered single bonds, with formal charges of −1 on both the oxygen and the nitrogen, and a formal charge of +2 on the sulfur.