
Square pyramidal molecular geometry
Shape of certain five-ligand chemical complexes / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Square pyramidal molecular geometry?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Square pyramidal geometry describes the shape of certain chemical compounds with the formula ML5 where L is a ligand. If the ligand atoms were connected, the resulting shape would be that of a pyramid with a square base. The point group symmetry involved is of type C4v. The geometry is common for certain main group compounds that have a stereochemically-active lone pair, as described by VSEPR theory. Certain compounds crystallize in both the trigonal bipyramidal and the square pyramidal structures, notably [Ni(CN)5]3−.[1]
Quick Facts Examples, Point group ...
| Square pyramidal molecular geometry | |
|---|---|
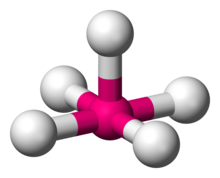 | |
| Examples | Chlorine pentafluoride (ClF5), MnCl2−5 |
| Point group | C4v |
| Coordination number | 5 |
Close
