
Sodium chloride
Chemical compound with formula NaCl / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sodium chloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
This article is about the chemical. For its familiar form, common table salt, see Salt. For the medical solutions, see Saline (medicine). For the mineral, see Halite.
"NaCl" redirects here. For other uses, see NaCl (disambiguation).
Sodium chloride /ˌsoʊdiəm ˈklɔːraɪd/,[8] commonly known as edible salt, is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chlorine ions. It is transparent or translucent, brittle, hygroscopic, and occurs as the mineral halite. In its edible form, it is commonly used as a condiment and food preservative. Large quantities of sodium chloride are used in many industrial processes, and it is a major source of sodium and chlorine compounds used as feedstocks for further chemical syntheses. Another major application of sodium chloride is deicing of roadways in sub-freezing weather.
Quick Facts Names, Identifiers ...
 Sodium chloride crystals in a form of halite | |
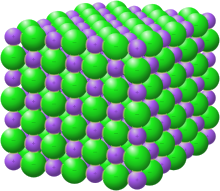 Crystal structure with sodium in purple and chloride in green[1] | |
| Names | |
|---|---|
| IUPAC name
Sodium chloride | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| 3534976 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.028.726 |
| EC Number |
|
| 13673 | |
| KEGG |
|
| MeSH | Sodium+chloride |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| NaCl | |
| Molar mass | 58.443 g/mol[2] |
| Appearance | Colorless cubic crystals[2] |
| Odor | Odorless |
| Density | 2.17 g/cm3[2] |
| Melting point | 800.7 °C (1,473.3 °F; 1,073.8 K)[2] |
| Boiling point | 1,465 °C (2,669 °F; 1,738 K)[2] |
| 360 g/L (25°C)[2] | |
| Solubility in ammonia | 21.5 g/L |
| Solubility in methanol | 14.9 g/L |
| −30.2·10−6 cm3/mol[3] | |
Refractive index (nD) |
1.5441 (at 589 nm)[4] |
| Structure[5] | |
| Face-centered cubic (see text), cF8 | |
| Fm3m (No. 225) | |
a = 564.02 pm | |
Formula units (Z) |
4 |
| octahedral at Na+ octahedral at Cl− | |
| Thermochemistry[6] | |
Heat capacity (C) |
50.5 J/(K·mol) |
Std molar entropy (S⦵298) |
72.10 J/(K·mol) |
Std enthalpy of formation (ΔfH⦵298) |
−411.120 kJ/mol |
| Pharmacology | |
| A12CA01 (WHO) B05CB01 (WHO), B05XA03 (WHO), S01XA03 (WHO) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
3 g/kg (oral, rats)[7] |
| Related compounds | |
Other anions |
Sodium fluoride Sodium bromide Sodium iodide Sodium astatide |
Other cations |
Lithium chloride Potassium chloride Rubidium chloride Caesium chloride Francium chloride |
| Supplementary data page | |
| Sodium chloride (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
