Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
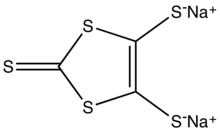
Sodium 1,3-dithiole-2-thione-4,5-dithiolate is the organosulfur compound with the formula Na2C3S5, abbreviated Na2dmit. It is the sodium salt of the conjugate base of the 4,5-bis(sulfanyl)-1,3-dithiole-2-thione. The salt is a precursor to dithiolene complexes and tetrathiafulvalenes.[1]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Disodium 2-sulfanylidene-2H-1,3-dithiole-4,5-bis(thiolate) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C3Na2S5 | |
| Molar mass | 242.31 g·mol−1 |
| Appearance | yellow solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Reduction of carbon disulfide with sodium affords sodium 1,3-dithiole-2-thione-4,5-dithiolate together with sodium trithiocarbonate:
Before the characterization of dmit2-, reduction of CS2 was thought to give tetrathiooxalate (Na2C2S4).[2]
The dianion C3S52- is purified as the tetraethylammonium salt of the zincate complex [Zn(C3S5)2]2-. This salt converts to the bis(thioester) upon treatment with benzoyl chloride:[3][1]
Cleavage of the thioester with sodium methoxide gives sodium 1,3-dithiole-2-thione-4,5-dithiolate:

Na2dmit undergoes S-alkylation.[5] Heating solutions of Na2dmit gives the isomeric 1,2-dithioledithiolate.
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.