
Quinalizarin
Chemical compound / From Wikipedia, the free encyclopedia
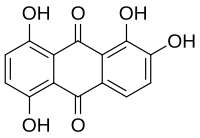
Quinalizarin or 1,2,5,8-tetrahydroxyanthraquinone is an organic compound with formula C
14H
8O
6. It is one of many tetrahydroxyanthraquinone isomers, formally derived from anthraquinone by replacement of four hydrogen atoms by hydroxyl (OH) groups at the 1, 2, 5, and 8 positions.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,2,5,8-Tetrahydroxyanthracene-9,10-dione | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.001.243 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C14H8O6 | |
| Molar mass | 272.212 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Quinalizarin is an inhibitor of the enzyme protein kinase CK2. It is more potent and selective than emodin.[1] It is also a potent catechol O-methyltransferase (COMT) inhibitor.[2][3]