
Prolintane
Stimulant and NDRI drug / From Wikipedia, the free encyclopedia
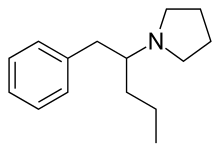
Prolintane (Catovit, Katovit, Promotil, Villescon) is a stimulant[2] and norepinephrine-dopamine reuptake inhibitor developed in the 1950s.[3] Being an amphetamine derivative, it is closely related in chemical structure to other drugs such as pyrovalerone, MDPV, and propylhexedrine and it has a similar mechanism of action.[4] Many cases of prolintane abuse have been reported.[5]
Quick Facts Clinical data, Routes ofadministration ...
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | oral, intranasal, rectal |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.007.077 |
| Chemical and physical data | |
| Formula | C15H23N |
| Molar mass | 217.356 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 133 °C (271 °F) |
| Boiling point | 153 °C (307 °F) |
| |
| |
| | |
Close
Under the trade-name "Katovit", prolintane was commercialized by the Spanish pharmaceutical company, FHER. Katovit was sold until 2001, and was most often used by students and workers as a stimulant to provide energy, promote alertness and concentration.[medical citation needed]