
Pregabalin
Anticonvulsant medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Pregabalin?
Summarize this article for a 10 year old
Pregabalin, sold under the brand name Lyrica among others, is an anticonvulsant, analgesic, and anxiolytic amino acid medication used to treat epilepsy, neuropathic pain, fibromyalgia, restless legs syndrome, opioid withdrawal, and generalized anxiety disorder (GAD).[13][17][18] Pregabalin also has antiallodynic properties.[19][20][21] Its use in epilepsy is as an add-on therapy for partial seizures.[13] It is a gabapentinoid medication (GABA analogue) which are drugs that are derivatives of γ-aminobutyric acid (GABA), an inhibitory neurotransmitter.[22][23][24][25] Pregabalin acts by inhibiting certain calcium channels.[13][26][27] When used before surgery, it reduces pain but results in greater sedation and visual disturbances.[28] It is taken by mouth.[13]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /priˈɡæbəlɪn/ |
| Trade names | Lyrica, others[1] |
| Other names | 3-isobutyl GABA, (S)-3-isobutyl-γ-aminobutyric acid |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605045 |
| License data |
|
| Pregnancy category | |
| Dependence liability | Physical: High[4] Psychological: Moderate[4] |
| Addiction liability | Low[4] (but varying with dosage and route of administration) |
| Routes of administration | By mouth |
| Drug class | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: High (≥90% rapidly absorbed; food has no significant effect on bioavailability)[11] |
| Protein binding | <1%[12] |
| Metabolites | N-methylpregabalin[11] |
| Onset of action | May occur within a week (pain)[13] |
| Elimination half-life | 4.5–7 hours[14] (mean 6.3 hours)[14][15] |
| Duration of action | Approx 8 to 12 hours [16] |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.119.513 |
| Chemical and physical data | |
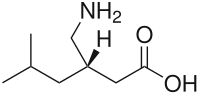
| Formula | C8H17NO2 |
| Molar mass | 159.229 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Common side effects include headache, dizziness, sleepiness, confusion, trouble with memory, poor coordination, dry mouth, problems with vision, and weight gain.[13][29] Serious side effects may include angioedema, drug misuse, and an increased suicide risk.[13] When pregabalin is taken at high doses over a long period of time, addiction may occur, but if taken at usual doses the risk is low.[4] Use during pregnancy or breastfeeding is of unclear safety.[30]
Pregabalin was approved for medical use in the United States in 2004.[13] It was developed as a successor to the related gabapentin.[31] It is available as a generic medication.[29][32][33][34][35] In 2021, it was the 73rd most commonly prescribed medication in the United States, with more than 8 million prescriptions.[36][37] In the US, pregabalin is a Schedule V controlled substance under the Controlled Substances Act of 1970,[13] which means that the drug has low abuse potential compared to substances in Schedules I-IV, however, there is still a potential for misuse.[38] Despite the low abuse potential, there have been reports of euphoria, improved happiness, excitement, calmness, and a "high" similar to marijuana with the use of pregabalin; there is a potential for developing dependence on these substances, and withdrawal symptoms may occur if the medication is abruptly discontinued.[39][40] It is a Class C controlled substance in the UK.[41] Therefore, pregabalin requires a prescription.[42][43] Furthermore, the prescription must clearly set forth the dose.[44] Pregabalin has potential for misuse. It can bring about an elevated mood in users. It can also have serious side effects, particularly when used in combination with other drugs.[44][45]