
Polyestradiol phosphate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Polyestradiol phosphate?
Summarize this article for a 10 year old
Polyestradiol phosphate (PEP), sold under the brand name Estradurin, is an estrogen medication which is used primarily in the treatment of prostate cancer in men.[1][9][2][10] It is also used in women to treat breast cancer, as a component of hormone therapy to treat low estrogen levels and menopausal symptoms, and as a component of feminizing hormone therapy for transgender women.[1][11] It is given by injection into muscle once every four weeks.[1][2][12]
 | |
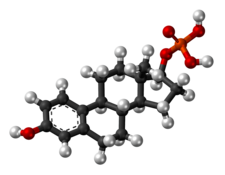 Skeletal structure of polyestradiol phosphate (top) and ball-and-stick model of estradiol phosphate (one monomer of polyestradiol phosphate) (bottom) | |
| Clinical data | |
|---|---|
| Trade names | Estradurin, Estradurine |
| Other names | PEP; Polymeric estradiol phosphate; Polymeric estradiol 17β-phosphate; Estradiol phosphate polymer; Estradiol 17β-phosphate polymer; Estradiol polymer with phosphoric acid; Leo-114 |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Intramuscular injection[1][2] |
| Drug class | Estrogen; Estrogen ester |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | IM: High |
| Protein binding | Estradiol: ~98% (to albumin and SHBGTooltip sex hormone-binding globulin)[3][4][1] |
| Metabolism | Mainly in the liver, to a lesser extent in the kidneys, gonads, and muscle (by phosphatases)[1] |
| Metabolites | Estradiol, phosphoric acid, and metabolites of estradiol[5][6] |
| Elimination half-life | PEP: 70 days (10 weeks)[7] Estradiol: 1–2 hours[8] |
| Excretion | Urine (as conjugates)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem SID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| Chemical and physical data | |
| Formula | (C18H23O4P)n (n = variable; n = 13) |
| Molar mass | Polymer: Variable Repeat unit: 334.347 g/mol |
| Melting point | 195 to 202 °C (383 to 396 °F) |
| (verify) | |
Common side effects of PEP include headache, breast tenderness, breast development, feminization, sexual dysfunction, infertility, and vaginal bleeding.[1][2] PEP is an estrogen and hence is an agonist of the estrogen receptor, the biological target of estrogens like estradiol.[2][6][5] It is an estrogen ester in the form of a polymer and is an extremely long-lasting prodrug of estradiol in the body.[2][12][5][6] The biological half-life of PEP is more than two months.[7] Because PEP works by being converted into estradiol, it is considered to be a natural and bioidentical form of estrogen.[5][2] The safety profile of parenteral estradiol esters like PEP is greatly improved relative to synthetic oral estrogens like ethinylestradiol and diethylstilbestrol.[2]
PEP was discovered around 1953 and was introduced for medical use in the United States in 1957.[12][13][14] Along with estradiol undecylate and estradiol valerate, it has been frequently used in the United States and Europe as a parenteral form of estrogen to treat men with prostate cancer.[15] However, it is no longer available in the United States.[13][16]