
Polycaprolactone
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Polycaprolactone?
Summarize this article for a 10 year old
Polycaprolactone (PCL) is a synthetic, semi-crystalline, biodegradable polyester with a melting point of about 60 °C and a glass transition temperature of about −60 °C.[2][3] The most common use of polycaprolactone is in the production of speciality polyurethanes. Polycaprolactones impart good resistance to water, oil, solvent and chlorine to the polyurethane produced.
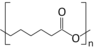 | |
| Names | |
|---|---|
| IUPAC name
(1,7)-Polyoxepan-2-one | |
| Systematic IUPAC name
Poly(hexano-6-lactone) | |
| Other names
2-Oxepanone homopolymer 6-Caprolactone polymer | |
| Identifiers | |
| Abbreviations | PCL |
| ChemSpider |
|
| Properties | |
| (C6H10O2)n | |
| Density | 1.145 g/cm3 |
| Melting point | 60 °C (140 °F) |
| Thermal conductivity | 0.05 W/(m K) @25°C[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

This polymer is often used as an additive for resins to improve their processing characteristics and their end use properties (e.g., impact resistance). Being compatible with a range of other materials, PCL can be mixed with starch to lower its cost and increase biodegradability or it can be added as a polymeric plasticizer to polyvinyl chloride (PVC).
Polycaprolactone is also used for splinting, modeling, and as a feedstock for prototyping systems such as fused filament fabrication 3D printers.