
Phenyllithium
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Phenyllithium?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Phenyllithium is an organometallic agent with the empirical formula C6H5Li. It is most commonly used as a metalating agent in organic syntheses and a substitute for Grignard reagents for introducing phenyl groups in organic syntheses.[3] Crystalline phenyllithium is colorless; however, solutions of phenyllithium are various shades of brown or red depending on the solvent used and the impurities present in the solute.[4]
Quick Facts Names, Identifiers ...
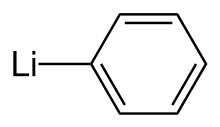 | |
 | |
| Names | |
|---|---|
| Systematic IUPAC name
Phenyllithium[1] | |
| Other names
Lithiobenzene[2] | |
| Identifiers | |
| |
3D model (JSmol) |
|
| Abbreviations | LiPh, PhLi |
| 506502 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.838 |
| EC Number |
|
| 2849 | |
| MeSH | phenyllithium |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| LiC 6H 5 | |
| Molar mass | 84.045 g mol−1 |
| Appearance | Colorless crystals |
| Density | 828 mg cm−3 |
| Boiling point | 140 to 143 °C (284 to 289 °F; 413 to 416 K) |
| Reacts | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
48.3-52.5 kJ mol−1 |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H226, H250, H261, H302, H312, H314, H332 | |
| P210, P222, P231+P232, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P280, P301+P312, P301+P330+P331, P302+P334, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P363, P370+P378, P402+P404, P403+P235, P405, P422, P501 | |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Related compounds |
phenylcopper, phenylsodium, phenylcobalt |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close