Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
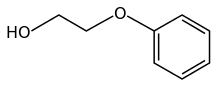
Phenoxyethanol is the organic compound with the formula C6H5OC2H4OH. It is a colorless oily liquid. It can be classified as a glycol ether and a phenol ether. It is a common preservative in vaccine formulations.[4]
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Phenoxyethan-1-ol | |
| Other names
Phenoxyethanol Ethylene glycol monophenyl ether Phenoxytolarosol Dowanol EP / EPH Protectol PE Emery 6705 Rose ether 1-Hydroxy-2-phenoxyethane β-hydroxyethyl phenyl ether Phenyl cellosolve Phenoxetol® | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.173 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H10O2 | |
| Molar mass | 138.166 g·mol−1 |
| Appearance | Colorless oily liquid |
| Odor | faint rose-like |
| Density | 1.102 g/cm3 |
| Melting point | −2 °C (28 °F; 271 K) |
| Boiling point | 247 °C (477 °F; 520 K) |
| 26 g/kg | |
| Solubility | Chloroform, Alkali, diethyl ether: soluble |
| Solubility in peanut oil | slightly |
| Solubility in olive oil | slightly |
| Solubility in acetone | miscible |
| Solubility in ethanol | miscible |
| Solubility in glycerol | miscible |
| Vapor pressure | 0.001 kPa (0.00015 psi) |
| Thermal conductivity | 0.169 W/(m⋅K) |
Refractive index (nD) |
1.534 (20 °C) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Harmful if swallowed Causes serious eye irritation |
| GHS labelling: | |
 | |
| Warning | |
| NFPA 704 (fire diamond) | |
| Flash point | 126 °C (259 °F; 399 K) |
| 430 °C (806 °F; 703 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
1850 mg/kg (rat, oral) |
| Related compounds | |
Related compounds |
phenetole |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenoxyethanol has germicidal and germistatic properties.[5] It is often used together with quaternary ammonium compounds.
Phenoxyethanol is used as a perfume fixative; an insect repellent; an antiseptic;[6] a solvent for cellulose acetate, dyes, inks, and resins; a preservative for pharmaceuticals, cosmetics and lubricants;[7] an anesthetic in fish aquaculture;[8][9] and in organic synthesis.
Phenoxyethanol is an alternative to formaldehyde-releasing preservatives.[10] In Japan and the European Union, its concentration in cosmetics is restricted to 1%.[11]
Phenoxyethanol is produced by the hydroxyethylation of phenol (Williamson synthesis), for example, in the presence of alkali-metal hydroxides or alkali-metal borohydrides. Phenoxyethanol has a long history of use, with its discovery credited to the German chemist Otto Schott in the early 20th century. Since then, it has been extensively studied and applied in various industries.[1]
Phenoxyethanol is effective against gram-negative and gram-positive bacteria, and the yeast Candida albicans.[12]
| Aromatic alcohol | Concentration (%) | Contact time (minutes) | |||
|---|---|---|---|---|---|
| Escherichia coli | Pseudomonas aeruginosa | Proteus mirabilis | Staphylococcus aureus | ||
| Benzyl alcohol | 1 | >30 | >30 | >30 | >30 |
| Phenethyl alcohol | 1.25 | 2.5 | 2.5 | 2.5 | >30 |
| 2.5 | 2.5 | 2.5 | 2.5 | 5 | |
| Phenoxyethanol | 1.25 | 15 | 2.5 | 2.5 | >30 |
| 2.5 | 2.5 | 2.5 | 2.5 | >30 | |
Phenoxyethanol is a vaccine preservative and potential allergen, which may result in a nodular reaction at the site of injection. Possible symptoms include rashes, eczema, and possible death.[14] It reversibly inhibits NMDAR-mediated ion currents.[15]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.