Periodic trends
Specific recurring patterns that are present in the modern periodic table / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Periodic trends?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
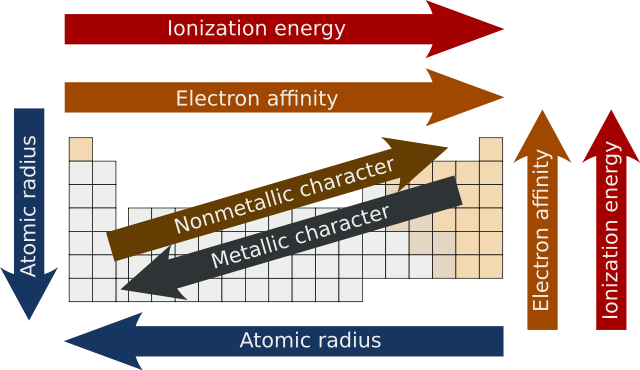
In chemistry, periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of certain elements when grouped by period and/or group. They were discovered by the Russian chemist Dmitri Mendeleev in 1863. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. These trends exist because of the similar electron configurations of the elements within their respective groups or periods; they reflect the periodic nature of the elements. These trends give a qualitative assessment of the properties of each element.[1][2]