
Pentene
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Pentene?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Pentenes (also called Pentylenes) are alkenes with the chemical formula C
5H
10. Each molecule contains one double bond within its molecular structure. Six different compounds are in this class, differing from each other by whether the carbon atoms are attached linearly or in a branched structure and whether the double bond has a cis or trans form.
Quick Facts Names, Identifiers ...
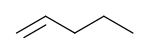 1-Pentene | |
 cis-2-Pentene | |
 trans-2-Pentene | |
| Names | |
|---|---|
| IUPAC names
Pent-1-ene cis-Pent-2-ene trans-Pent-2-ene | |
| Other names
amylene, n-amylene, n-pentene, beta-n-amylene, sym-methylethylethylene, pentylene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.042.636 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C5H10 | |
| Molar mass | 70.135 g·mol−1 |
| Density | 0.64 g/cm3 (1-pentene)[1] |
| Melting point | −165.2 °C (−265.4 °F; 108.0 K) (1-pentene)[1] |
| Boiling point | 30 °C (86 °F; 303 K) (1-pentene)[1] |
| -53.7·10−6 cm3/mol | |
| Hazards | |
| Safety data sheet (SDS) | MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close