
Parecoxib
Type of nonsteroidal anti-inflammatory drug / From Wikipedia, the free encyclopedia
For the "dynastat" type of hybrid airship, see Hybrid airship.
Parecoxib, sold under the brand name Dynastat among others, is a water-soluble and injectable prodrug of valdecoxib. Parecoxib is a COX2 selective inhibitor. It is injectable. It is approved in the European Union for short term perioperative pain control.
Quick Facts Clinical data, AHFS/Drugs.com ...
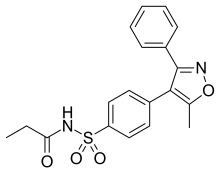 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Intravenous, intramuscular |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% |
| Protein binding | 98% |
| Metabolism | Liver to valdecoxib and propionic acid CYP extensively involved (mainly CYP3A4 and 2C9) |
| Elimination half-life | 22 minutes (parecoxib) 8 hours (valdecoxib) |
| Excretion | Kidney (70%, metabolites) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.230.078 |
| Chemical and physical data | |
| Formula | C19H18N2O4S |
| Molar mass | 370.42 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Close
It was patented in 1996 and approved for medical use in 2002.[3]