
Nesiritide
Pharmaceutical drug / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Nesiritide?
Summarize this article for a 10 year old
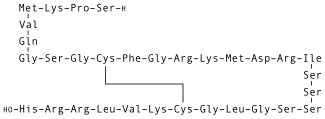
Nesiritide, sold under the brand name Natrecor, is the recombinant form of the 32 amino acid human B-type natriuretic peptide, which is normally produced by the ventricular myocardium. Nesiritide works to facilitate cardiovascular fluid homeostasis through counterregulation of the renin–angiotensin–aldosterone system, stimulating cyclic guanosine monophosphate, leading to smooth muscle cell relaxation.
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Routes of administration | IV only |
| ATC code | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C143H244N50O42S4 |
| Molar mass | 3464.07 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Nesiritide was believed initially to be beneficial for acute decompensated congestive heart failure. It received approval from the United States' Food and Drug Administration for this purpose in 2001 after initial non-approval. In July 2011, the results of the largest study so far for nesiritide was published in The New England Journal of Medicine. The study failed to show a difference between nesiritide and placebo on mortality or re-hospitalizations.[1]